Molecular identification of rapidly adapting mechanoreceptors and their developmental dependence on ret signaling
- PMID: 20064391
- PMCID: PMC2813518
- DOI: 10.1016/j.neuron.2009.11.003
Molecular identification of rapidly adapting mechanoreceptors and their developmental dependence on ret signaling
Abstract
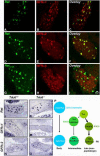
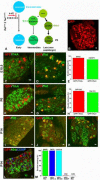
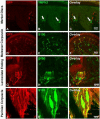
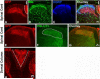
In mammals, the first step in the perception of form and texture is the activation of trigeminal or dorsal root ganglion (DRG) mechanosensory neurons, which are classified as either rapidly (RA) or slowly adapting (SA) according to their rates of adaptation to sustained stimuli. The molecular identities and mechanisms of development of RA and SA mechanoreceptors are largely unknown. We found that the "early Ret(+)" DRG neurons are RA mechanoreceptors, which form Meissner corpuscles, Pacinian corpuscles, and longitudinal lanceolate endings. The central projections of these RA mechanoreceptors innervate layers III through V of the spinal cord and terminate within discrete subdomains of the dorsal column nuclei. Moreover, mice lacking Ret signaling components are devoid of Pacinian corpuscles and exhibit a dramatic disruption of RA mechanoreceptor projections to both the spinal cord and medulla. Thus, the early Ret(+) neurons are RA mechanoreceptors and Ret signaling is required for the assembly of neural circuits underlying touch perception.
2009 Elsevier Inc. All rights reserved.
Figures
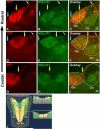
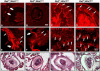
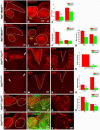
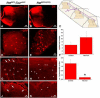
Similar articles
-
RETouching upon mechanoreceptors.Neuron. 2009 Dec 24;64(6):773-6. doi: 10.1016/j.neuron.2009.12.014. Neuron. 2009. PMID: 20064382 Free PMC article.
-
Low-threshold mechanoreceptor subtypes selectively express MafA and are specified by Ret signaling.Neuron. 2009 Dec 24;64(6):857-70. doi: 10.1016/j.neuron.2009.12.004. Neuron. 2009. PMID: 20064392
-
c-Maf is required for the development of dorsal horn laminae III/IV neurons and mechanoreceptive DRG axon projections.J Neurosci. 2012 Apr 18;32(16):5362-73. doi: 10.1523/JNEUROSCI.6239-11.2012. J Neurosci. 2012. PMID: 22514301 Free PMC article.
-
Immunohistochemistry of human cutaneous Meissner and pacinian corpuscles.Microsc Res Tech. 1996 Jul 1;34(4):351-61. doi: 10.1002/(SICI)1097-0029(19960701)34:4<351::AID-JEMT6>3.0.CO;2-R. Microsc Res Tech. 1996. PMID: 8807618 Review.
-
The Meissner and Pacinian sensory corpuscles revisited new data from the last decade.Microsc Res Tech. 2009 Apr;72(4):299-309. doi: 10.1002/jemt.20651. Microsc Res Tech. 2009. PMID: 19012318 Review.
Cited by
-
The development of somatosensory neurons: Insights into pain and itch.Curr Top Dev Biol. 2021;142:443-475. doi: 10.1016/bs.ctdb.2020.10.005. Epub 2020 Nov 5. Curr Top Dev Biol. 2021. PMID: 33706924 Free PMC article. Review.
-
Biphasic functions for the GDNF-Ret signaling pathway in chemosensory neuron development and diversification.Proc Natl Acad Sci U S A. 2018 Jan 16;115(3):E516-E525. doi: 10.1073/pnas.1708838115. Epub 2017 Dec 27. Proc Natl Acad Sci U S A. 2018. PMID: 29282324 Free PMC article.
-
Genetic Tracing of Cav3.2 T-Type Calcium Channel Expression in the Peripheral Nervous System.Front Mol Neurosci. 2017 Mar 15;10:70. doi: 10.3389/fnmol.2017.00070. eCollection 2017. Front Mol Neurosci. 2017. PMID: 28360836 Free PMC article.
-
Characterization of retinal ganglion cell, horizontal cell, and amacrine cell types expressing the neurotrophic receptor tyrosine kinase Ret.J Comp Neurol. 2018 Mar 1;526(4):742-766. doi: 10.1002/cne.24367. Epub 2017 Dec 19. J Comp Neurol. 2018. PMID: 29218725 Free PMC article.
-
Distinct Modes of Presynaptic Inhibition of Cutaneous Afferents and Their Functions in Behavior.Neuron. 2019 Apr 17;102(2):420-434.e8. doi: 10.1016/j.neuron.2019.02.002. Epub 2019 Feb 27. Neuron. 2019. PMID: 30826183 Free PMC article.
References
-
- Airaksinen MS, Koltzenburg M, Lewin GR, Masu Y, Helbig C, Wolf E, Brem G, Toyka KV, Thoenen H, Meyer M. Specific subtypes of cutaneous mechanoreceptors require neurotrophin-3 following peripheral target innervation. Neuron. 1996;16:287–295. - PubMed
-
- Airaksinen MS, Saarma M. The GDNF family: signalling, biological functions and therapeutic value. Nat Rev Neurosci. 2002;3:383–394. - PubMed
-
- Airaksinen MS, Titievsky A, Saarma M. GDNF family neurotrophic factor signaling: four masters, one servant? Mol Cell Neurosci. 1999;13:313–325. - PubMed
-
- Albrecht F. L. R. a. P. J. The Senses: A Comprehensive Reference. Academic Press; San Diego: 2008. Cutaneous Mechanisms of Tactile Perception: Morphological and Chemical Organization of the Innervation to the Skin; pp. 1–32.
-
- Brown AG. Organization in the spinal cord. Heidelberg; Springer-Verlag; Berlin: New York: 1981.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

