Mitochondrial DNA toxicity in forebrain neurons causes apoptosis, neurodegeneration, and impaired behavior
- PMID: 20065039
- PMCID: PMC2832488
- DOI: 10.1128/MCB.01149-09
Mitochondrial DNA toxicity in forebrain neurons causes apoptosis, neurodegeneration, and impaired behavior
Abstract
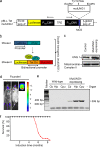
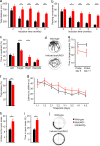
Mitochondrial dysfunction underlying changes in neurodegenerative diseases is often associated with apoptosis and a progressive loss of neurons, and damage to the mitochondrial genome is proposed to be involved in such pathologies. In the present study we designed a mouse model that allows us to specifically induce mitochondrial DNA toxicity in the forebrain neurons of adult mice. This is achieved by CaMKIIalpha-regulated inducible expression of a mutated version of the mitochondrial UNG DNA repair enzyme (mutUNG1). This enzyme is capable of removing thymine from the mitochondrial genome. We demonstrate that a continual generation of apyrimidinic sites causes apoptosis and neuronal death. These defects are associated with behavioral alterations characterized by increased locomotor activity, impaired cognitive abilities, and lack of anxietylike responses. In summary, whereas mitochondrial base substitution and deletions previously have been shown to correlate with premature and natural aging, respectively, we show that a high level of apyrimidinic sites lead to mitochondrial DNA cytotoxicity, which causes apoptosis, followed by neurodegeneration.
Figures




References
-
- Anderson, M. F., and N. R. Sims. 2000. Improved recovery of highly enriched mitochondrial fractions from small brain tissue samples. Brain Res. Brain Res. Protoc. 5:95-101. - PubMed
-
- Baumgartel, K., D. Genoux, H. Welzl, R. Y. Tweedie-Cullen, K. Koshibu, M. Livingstone-Zatchej, C. Mamie, and I. M. Mansuy. 2008. Control of the establishment of aversive memory by calcineurin and Zif268. Nat. Neurosci. 11:572-578. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
