Induction of B-cell immune tolerance by antigen-modified cytotoxic T lymphocytes
- PMID: 20065917
- PMCID: PMC2844488
- DOI: 10.1097/TP.0b013e3181ca9048
Induction of B-cell immune tolerance by antigen-modified cytotoxic T lymphocytes
Abstract
Background: Third-party-specific cytotoxic T lymphocytes (CTL), or veto CTL, are being assessed as a cellular therapeutic for the induction of T-cell tolerance during transplantation. Conceptually, veto cell-expressed antigens (Ags) may induce B-cell immune responses, and this may have deleterious consequences. Whether veto cells induce immunity, tolerance, or are ignored by B lymphocytes has, however, not been addressed.
Methods: CTL were retrovirally transduced with a model cell surface Ag to generate veto CTL. The impact of CTL-specific Ag expression on the activation and tolerization of Ag-specific B cells was assessed in vitro and, using adoptive transfer models, in vivo.
Results: In vitro, CTL-expressed Ag induced an abortive proliferative response in specific B lymphocytes, whereby an initial proliferative burst was followed by cell death. In vivo, the administration of veto CTL also induced B-cell tolerance. Specific immunoglobulin was not detected after subsequent immunization with a veto cell-expressed Ag. Modeling of this effect with Ag-specific B-cell receptor transgenic B lymphocytes demonstrated that Ag-specific B cells were eliminated by the veto CTL; the cell division was accompanied by the exhaustion and depletion of responding cells. Veto-induced B-cell tolerance could be wholly abrogated by treatment with the toll-like receptor ligand lipopolysaccharide, implying that this tolerance resulted from the absence of adequate supplemental signals during antigenic stimulation.
Conclusions: Veto CTL are effective promoters of B-cell tolerance. Further assessment of their therapeutic potential in this regard is warranted.
Conflict of interest statement
The authors have no commercial interests relating to this work.
Figures
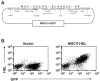
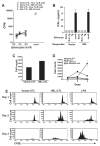
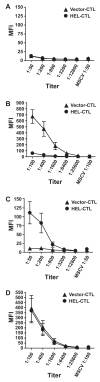
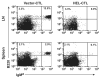

Comment in
-
Are B cells agreeable to veto?Transplantation. 2010 Mar 27;89(6):646-7. doi: 10.1097/TP.0b013e3181ca9065. Transplantation. 2010. PMID: 20048694 No abstract available.
Similar articles
-
Are B cells agreeable to veto?Transplantation. 2010 Mar 27;89(6):646-7. doi: 10.1097/TP.0b013e3181ca9065. Transplantation. 2010. PMID: 20048694 No abstract available.
-
Induction of tolerance and immunity by redirected B cell-specific cytolytic T lymphocytes.Gene Ther. 2007 Dec;14(24):1739-49. doi: 10.1038/sj.gt.3303045. Epub 2007 Oct 11. Gene Ther. 2007. PMID: 17928872
-
Studies on the mechanism of suppression of primary cytotoxic responses by cloned cytotoxic T lymphocytes.J Immunol. 1984 Oct;133(4):1769-74. J Immunol. 1984. PMID: 6236258
-
Veto function in vitro and in vivo.Int Rev Immunol. 1989 May;4(2):175-91. doi: 10.3109/08830188909044780. Int Rev Immunol. 1989. PMID: 2485841 Review.
-
Veto suppression: the peripheral way of T cell tolerization.Exp Clin Immunogenet. 1993;10(4):179-88. Exp Clin Immunogenet. 1993. PMID: 7907886 Review.
Cited by
-
The use of donor-derived veto cells in hematopoietic stem cell transplantation.Front Immunol. 2012 May 2;3:93. doi: 10.3389/fimmu.2012.00093. eCollection 2012. Front Immunol. 2012. PMID: 22566971 Free PMC article.
-
Modulation of CD8+ T cell responses to AAV vectors with IgG-derived MHC class II epitopes.Mol Ther. 2013 Sep;21(9):1727-37. doi: 10.1038/mt.2013.166. Epub 2013 Jul 16. Mol Ther. 2013. PMID: 23857231 Free PMC article.
-
Altered differentiation, diminished pathogenicity, and regulatory activity of myelin-specific T cells expressing an enhanced affinity TCR.J Immunol. 2011 Dec 1;187(11):5521-31. doi: 10.4049/jimmunol.1102202. Epub 2011 Oct 24. J Immunol. 2011. PMID: 22025553 Free PMC article.
-
Siglecs induce tolerance to cell surface antigens by BIM-dependent deletion of the antigen-reactive B cells.J Immunol. 2014 Nov 1;193(9):4312-21. doi: 10.4049/jimmunol.1401723. Epub 2014 Sep 24. J Immunol. 2014. PMID: 25252961 Free PMC article.
References
-
- Soiffer R. Immune modulation and chronic graft-versus-host disease. Bone Marrow Transplant. 2008;42 1:S66–S69. - PubMed
-
- Zarkhin V, Li L, Sarwal M. “To B or not to B?” B-cells and graft rejection. Transplantation. 2008;85:1705–1714. - PubMed
-
- Brennan DC, Mohanakumar T, Flye MW. Donor-specific transfusion and donor bone marrow infusion in renal transplantation tolerance: a review of efficacy and mechanisms. Am J Kidney Dis. 1995;26:701–715. - PubMed
-
- Fabre JW, Morris PJ. The effect of donor strain blood pretreatment on renal allograft rejection in rats. Transplantation. 1972;14:608–617. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

