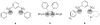Novel bifunctional sulfonamides catalyze an enantioselective conjugate addition
- PMID: 20066026
- PMCID: PMC2659672
- DOI: 10.1016/j.tet.2008.11.027
Novel bifunctional sulfonamides catalyze an enantioselective conjugate addition
Abstract
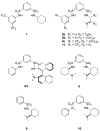
A new bifunctional organocatalyst with a novel structural and functional motif has been developed. This bifunctional sulfonamide organocatalyst was used in the conjugate addition of 1,3-dicarbonyl compounds (13) to β-nitrostyrenes (12). Yields up to 91% and enantiomeric excesses up to 79% were obtained in this reaction. This catalyst activates both 13 via its basic moiety and 12 through hydrogen bonding.
Figures
Similar articles
-
New bifunctional 1,3-diamine organocatalysts derived from (+)-camphoric acid for asymmetric Michael addition of 1,3-dicarbonyl compounds to nitroolefins.Chirality. 2022 May;34(5):782-795. doi: 10.1002/chir.23424. Epub 2022 Feb 15. Chirality. 2022. PMID: 35166402
-
The synthesis of chiral β-naphthyl-β-sulfanyl ketones via enantioselective sulfa-Michael reaction in the presence of a bifunctional cinchona/sulfonamide organocatalyst.Beilstein J Org Chem. 2021 Feb 18;17:494-503. doi: 10.3762/bjoc.17.43. eCollection 2021. Beilstein J Org Chem. 2021. PMID: 33727972 Free PMC article.
-
Asymmetric synthesis of highly functionalized tetrahydropyrans via a one-pot organocatalytic Michael/Henry/ketalization sequence.Org Lett. 2014 Jul 18;16(14):3636-9. doi: 10.1021/ol501236a. Epub 2014 Jun 27. Org Lett. 2014. PMID: 24971998 Free PMC article.
-
Synthesis and characterization of binary-complex models of ureas and 1,3-dicarbonyl compounds: deeper insights into reaction mechanisms using snap-shot structural analysis.J Org Chem. 2014 Feb 21;79(4):1805-17. doi: 10.1021/jo4028775. Epub 2014 Feb 4. J Org Chem. 2014. PMID: 24450387
-
[Development of Green Asymmetric Organocatalytic Synthesis].Yakugaku Zasshi. 2021;141(10):1137-1145. doi: 10.1248/yakushi.21-00130. Yakugaku Zasshi. 2021. PMID: 34602510 Review. Japanese.
Cited by
-
Chiral Thioureas-Preparation and Significance in Asymmetric Synthesis and Medicinal Chemistry.Molecules. 2020 Jan 18;25(2):401. doi: 10.3390/molecules25020401. Molecules. 2020. PMID: 31963671 Free PMC article. Review.
-
Exploring the potential of diarylacetylenediols as hydrogen bonding catalysts.J Org Chem. 2013 Sep 6;78(17):8340-53. doi: 10.1021/jo400926n. Epub 2013 Aug 14. J Org Chem. 2013. PMID: 23869597 Free PMC article.
References
-
-
For recent general reviews, see for example: (a) Enders D, Narine AA. J Org Chem. 2008;73:7857–7870.Dondoni A, Massi A. Angew Chem Int Ed. 2008;47:4638–4660.Melchiorre P, Marigo M, Carlone A, Bartoli G. Angew Chem Int Ed. 2008;47:6138–6171.
-
-
-
For recent reviews, see for example: (a) Doyle AG, Jacobsen EN. Chem Rev. 2007;107:5713–5743.Akiyama T. Chem Rev. 2007;107:5744–5758.
-
-
-
For thioureas derived from 1,2-cyclohexanediamine, see for example: (a) Okino T, Hoashi Y, Takemoto Y. J Am Chem Soc. 2003;125:12672–12673.Berkessel A, Cleemann F, Mukherjee S, Müller TN, Lex J. Angew Chem Int Ed. 2005;44:807–811.Fuerst DE, Jacobsen EN. J Am Chem Soc. 2005;127:8964–8965.Dove AP, Pratt RC, Lohmeijer BGG, Waymouth RM, Hedrick JL. J Am Chem Soc. 2005;127:13798–13799.Li H, Wang J, Zu L, Wang W. Tetrahedron Lett. 2006;47:2585–2589.Liu TY, Li R, Chai Q, Long J, Li BJ, Wu Y, Ding LS, Chen YC. Chem Eur J. 2006;13:319–327.
-
-
-
For thioureas derived from 1,2-diphenylethylenediamine, see for example: (a) Okino T, Hoashi Y, Furukawa T, Xu X, Takemoto Y. J Am Chem Soc. 2005;127:119–125.Liu TY, Long J, Li BJ, Jiang L, Li R, Wu Y, Ding LS, Chen YC. Org Biomol Chem. 2006;4:2097–2099.
-
-
-
For thioureas derived from BINAP, see for example: (a) Wang J, Li H, Yu X, Zu L, Wang W. Org Lett. 2005;7:4293–4296.Wang J, Li H, Duan W, Zu L, Wang W. Org Lett. 2005;7:4713–4716.
-
Grants and funding
LinkOut - more resources
Full Text Sources