Structural studies of bacterioferritin B from Pseudomonas aeruginosa suggest a gating mechanism for iron uptake via the ferroxidase center
- PMID: 20067302
- PMCID: PMC2852880
- DOI: 10.1021/bi9015204
Structural studies of bacterioferritin B from Pseudomonas aeruginosa suggest a gating mechanism for iron uptake via the ferroxidase center
Abstract
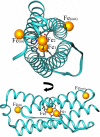
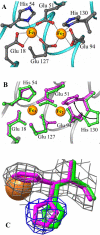
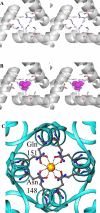
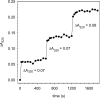
The structure of recombinant Pseudomonas aeruginosa bacterioferritin B (Pa BfrB) has been determined from crystals grown from protein devoid of core mineral iron (as-isolated) and from protein mineralized with approximately 600 iron atoms (mineralized). Structures were also obtained from crystals grown from mineralized BfrB after they had been soaked in an FeSO(4) solution (Fe soak) and in separate experiments after they had been soaked in an FeSO(4) solution followed by a soak in a crystallization solution (double soak). Although the structures consist of a typical bacterioferritin fold comprised of a nearly spherical 24-mer assembly that binds 12 heme molecules, comparison of microenvironments observed in the distinct structures provided interesting insights. The ferroxidase center in the as-isolated, mineralized, and double-soak structures is empty. The ferroxidase ligands (except His130) are poised to bind iron with minimal conformational changes. The His130 side chain, on the other hand, must rotate toward the ferroxidase center to coordinate iron. In comparison, the structure obtained from crystals soaked in an FeSO(4) solution displays a fully occupied ferroxidase center and iron bound to the internal, Fe((in)), and external, Fe((out)), surfaces of Pa BfrB. The conformation of His130 in this structure is rotated toward the ferroxidase center and coordinates an iron ion. The structures also revealed a pore on the surface of Pa BfrB that likely serves as a port of entry for Fe(2+) to the ferroxidase center. On its opposite end, the pore is capped by the side chain of His130 when it adopts its "gate-closed" conformation that enables coordination to a ferroxidase iron. A change to its "gate-open", noncoordinative conformation creates a path for the translocation of iron from the ferroxidase center to the interior cavity. These structural observations, together with findings obtained from iron incorporation measurements in solution, suggest that the ferroxidase pore is the dominant entry route for the uptake of iron by Pa BfrB. These findings, which are clearly distinct from those made with Escherichia coli Bfr [Crow, A. C., Lawson, T. L., Lewin, A., Moore, G. R., and Le Brun, N. E. (2009) J. Am. Chem. Soc. 131, 6808-6813], indicate that not all bacterioferritins operate in the same manner.
Figures




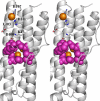

Similar articles
-
Two distinct ferritin-like molecules in Pseudomonas aeruginosa: the product of the bfrA gene is a bacterial ferritin (FtnA) and not a bacterioferritin (Bfr).Biochemistry. 2011 Jun 14;50(23):5236-48. doi: 10.1021/bi2004119. Epub 2011 May 20. Biochemistry. 2011. PMID: 21574546 Free PMC article.
-
Concerted motions networking pores and distant ferroxidase centers enable bacterioferritin function and iron traffic.Biochemistry. 2015 Mar 3;54(8):1611-27. doi: 10.1021/bi501255r. Epub 2015 Feb 17. Biochemistry. 2015. PMID: 25640193 Free PMC article.
-
Bacterioferritin: Structure, Dynamics, and Protein-Protein Interactions at Play in Iron Storage and Mobilization.Acc Chem Res. 2017 Feb 21;50(2):331-340. doi: 10.1021/acs.accounts.6b00514. Epub 2017 Feb 8. Acc Chem Res. 2017. PMID: 28177216 Free PMC article.
-
Iron storage in bacteria.Adv Microb Physiol. 1998;40:281-351. doi: 10.1016/s0065-2911(08)60134-4. Adv Microb Physiol. 1998. PMID: 9889981 Review.
-
Ferritins, iron uptake and storage from the bacterioferritin viewpoint.EMBO J. 2003 May 1;22(9):1959-68. doi: 10.1093/emboj/cdg215. EMBO J. 2003. PMID: 12727864 Free PMC article. Review.
Cited by
-
Protein dynamics and ion traffic in bacterioferritin.Biochemistry. 2012 Dec 11;51(49):9900-10. doi: 10.1021/bi3013388. Epub 2012 Nov 30. Biochemistry. 2012. PMID: 23167635 Free PMC article.
-
Pseudomonas aeruginosa gene PA4880 encodes a Dps-like protein with a Dps fold, bacterioferritin-type ferroxidase centers, and endonuclease activity.Front Mol Biosci. 2024 May 22;11:1390745. doi: 10.3389/fmolb.2024.1390745. eCollection 2024. Front Mol Biosci. 2024. PMID: 38841187 Free PMC article.
-
Two distinct ferritin-like molecules in Pseudomonas aeruginosa: the product of the bfrA gene is a bacterial ferritin (FtnA) and not a bacterioferritin (Bfr).Biochemistry. 2011 Jun 14;50(23):5236-48. doi: 10.1021/bi2004119. Epub 2011 May 20. Biochemistry. 2011. PMID: 21574546 Free PMC article.
-
Simulations of stressosome activation emphasize allosteric interactions between RsbR and RsbT.BMC Syst Biol. 2013 Jan 15;7:3. doi: 10.1186/1752-0509-7-3. BMC Syst Biol. 2013. PMID: 23320651 Free PMC article.
-
The FinR-regulated essential gene fprA, encoding ferredoxin NADP+ reductase: Roles in superoxide-mediated stress protection and virulence of Pseudomonas aeruginosa.PLoS One. 2017 Feb 10;12(2):e0172071. doi: 10.1371/journal.pone.0172071. eCollection 2017. PLoS One. 2017. PMID: 28187184 Free PMC article.
References
-
- Yang X, Le Brun NE, Thomson AJ, Moore GR, Chasteen ND. The Iron Oxidation and Hydrolysis Chemistry of Escherichia coli Bacterioferritin. Biochemistry. 2000;39:4915–4923. - PubMed
-
- Bunker J, Lowry T, Davis G, Zhang B, Brosnahan D, Lindsay S, Costen R, Choi S, Arosio P, Watt GD. Kinetic Studies of Iron Deposition Catalyzed by Recombinant Human Liver Heavy and Light Ferritins and Azotobacter vinelandii Bacterioferritin using O2 and H2O2 as Oxidants. Biophys. Chem. 2005;114:235–244. - PubMed
-
- Zhao G, Bou-Abdallah F, Arosio P, Levi S, Janus-Chandler C, Chasteen ND. Multiple Pathways for Mineral Core Formation in Mammalian Apoferritin. The Role of Hydrogen Peroxide. Biochemistry. 2003;42:3142–3150. - PubMed
-
- Lewin A, Moore GR, Le Brun NE. Formation of Protein-Coated Iron Minerals. Dalton Trans. 2005:3597–3610. - PubMed
-
- Liu X, Theil EC. Ferritins: Dynamic Management of Biological Iron and Oxygen Chemistry. Acc. Chem. Res. 2005;38:167–175. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

