Identification of circulating neuropilin-1 and dose-dependent elevation following anti-neuropilin-1 antibody administration
- PMID: 20068394
- PMCID: PMC2726604
- DOI: 10.4161/mabs.1.4.8885
Identification of circulating neuropilin-1 and dose-dependent elevation following anti-neuropilin-1 antibody administration
Abstract
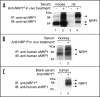
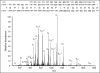
Neuropilin-1 (NRP1) acts as a co-receptor for class 3 semaphorins and vascular endothelial growth factor and is an attractive angiogenesis target for cancer therapy. In addition to the transmembrane form, naturally occurring soluble NRP1 proteins containing part of the extracellular domain have been identified in tissues and a cell line. We developed ELISAs to study the existence of circulating NRP1 and to quantify it in serum. As measured by ELISAs, circulating NRP1 levels in mice, rats, monkeys and humans were 427 +/- 77, 20 +/- 3, 288 +/- 86 and 322 +/- 82 ng/ml (mean +/- standard deviation; n > or = 10), respectively. Anti-NRP1(B), a human monoclonal antibody, has been selected from a synthetic phage library. A 4-fold increase in circulating NRP1 was observed in mice receiving a single dose of 10 mg/kg anti-NRP1(B) antibody. In rats and monkeys receiving single injections of anti-NRP1(B) at different dose levels, higher doses of antibody resulted in greater and more prolonged increases in circulating NRP1. Maximum increases were 56- and 7-fold for rats and monkeys receiving 50 mg/kg anti-NRP1(B), respectively. In addition to the soluble NRP1 isoforms, for the first time, a approximately 120 kDa circulating NRP1 protein containing the complete extracellular domain was detected in serum by western blot and mass spectrometry analysis. This protein increased more than the putative soluble NRP1 bands in anti-NRP1(B) treated mouse, rat and monkey sera compared with untreated controls, suggesting that anti-NRP1(B) induced membrane NRP1 shedding.
Figures

Similar articles
-
Quantitation of Circulating Neuropilin-1 in Human, Monkey, Mouse, and Rat Sera by ELISA.Methods Mol Biol. 2015;1332:39-48. doi: 10.1007/978-1-4939-2917-7_3. Methods Mol Biol. 2015. PMID: 26285744
-
Identification of a natural soluble neuropilin-1 that binds vascular endothelial growth factor: In vivo expression and antitumor activity.Proc Natl Acad Sci U S A. 2000 Mar 14;97(6):2573-8. doi: 10.1073/pnas.040337597. Proc Natl Acad Sci U S A. 2000. PMID: 10688880 Free PMC article.
-
Neuropilin-1 expression in cancer and development.J Pathol. 2012 Jan;226(1):50-60. doi: 10.1002/path.2989. Epub 2011 Oct 25. J Pathol. 2012. PMID: 22025255
-
Neuropilin 1: function and therapeutic potential in cancer.Cancer Immunol Immunother. 2014 Feb;63(2):81-99. doi: 10.1007/s00262-013-1500-0. Epub 2013 Nov 22. Cancer Immunol Immunother. 2014. PMID: 24263240 Free PMC article. Review.
-
Neuropilin-1 enforces extracellular matrix signalling via ABL1 to promote angiogenesis.Biochem Soc Trans. 2014 Oct;42(5):1429-34. doi: 10.1042/BST20140141. Biochem Soc Trans. 2014. PMID: 25233427 Review.
Cited by
-
MiR-200c sensitizes Olaparib-resistant ovarian cancer cells by targeting Neuropilin 1.J Exp Clin Cancer Res. 2020 Jan 2;39(1):3. doi: 10.1186/s13046-019-1490-7. J Exp Clin Cancer Res. 2020. PMID: 31898520 Free PMC article.
-
Characterisation of the interaction of neuropilin-1 with heparin and a heparan sulfate mimetic library of heparin-derived sugars.PeerJ. 2014 Jun 26;2:e461. doi: 10.7717/peerj.461. eCollection 2014. PeerJ. 2014. PMID: 25024924 Free PMC article.
-
Targeting neuropilins as a viable SARS-CoV-2 treatment.FEBS J. 2021 Sep;288(17):5122-5129. doi: 10.1111/febs.16096. Epub 2021 Jul 12. FEBS J. 2021. PMID: 34185437 Free PMC article. Review.
-
Soluble Neuropilin-1 is an independent marker of poor prognosis in early breast cancer.J Cancer Res Clin Oncol. 2021 Aug;147(8):2233-2238. doi: 10.1007/s00432-021-03635-1. Epub 2021 Apr 21. J Cancer Res Clin Oncol. 2021. PMID: 33884469 Free PMC article.
-
The roles of Eph receptors, neuropilin-1, P2X7, and CD147 in COVID-19-associated neurodegenerative diseases: inflammasome and JaK inhibitors as potential promising therapies.Cell Mol Biol Lett. 2022 Feb 2;27(1):10. doi: 10.1186/s11658-022-00311-1. Cell Mol Biol Lett. 2022. PMID: 35109786 Free PMC article. Review.
References
-
- He Z, Tessier-Lavigne M. Neuropilin is a receptor for the axonal chemorepellent Semaphorin III. Cell. 1997;90:739–751. - PubMed
-
- Kolodkin AL, Levengood DV, Rowe EG, Tai YT, Giger RJ, Ginty DD. Neuropilin is a semaphorin III receptor. Cell. 1997;90:753–762. - PubMed
-
- Soker S, Takashima S, Miao HQ, Neufeld G, Klagsbrun M. Neuropilin-1 is expressed by endothelial and tumor cells as an isoform-specific receptor for vascular endothelial growth factor. Cell. 1998;92:735–745. - PubMed
-
- Kitsukawa T, Shimono A, Kawakami A, Kondoh H, Fujisawa H. Overexpression of a membrane protein, neuropilin, in chimeric mice causes anomalies in the cardiovascular system, nervous system and limbs. Development. 1995;121:4309–4318. - PubMed
-
- Kawasaki T, Kitsukawa T, Bekku Y, Matsuda Y, Sanbo M, Yagi T, et al. A requirement for neuropilin-1 in embryonic vessel formation. Development. 1999;126:4895–4902. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
