Predictors of post-treatment relapse to smoking in successful quitters: pooled data from two phase III varenicline trials
- PMID: 20071105
- PMCID: PMC2875368
- DOI: 10.1016/j.drugalcdep.2009.12.022
Predictors of post-treatment relapse to smoking in successful quitters: pooled data from two phase III varenicline trials
Abstract
Background: Identifying predictors of smoking relapse helps to elucidate the challenges of long-term smoking cessation and provides direction for improved treatment development.
Methods: In this post hoc data analysis, we examined predictors of relapse from end-of-treatment (week 13) through 1-year follow-up (week 52) for treatment-responding participants who achieved the primary efficacy endpoint of 4-week continuous abstinence (weeks 9-12), during two phase III varenicline trials.
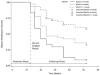
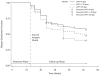
Results: Of 626 smokers classified as treatment responders for all treatment groups across both trials, 301 (48%) relapsed during follow-up (weeks 13-52). The odds of relapsing were almost 5 times greater (odds ratio [OR]=4.92, 95% confidence interval [CI]: 2.77-8.97; p<.001) for treatment responders who did not initiate continuous abstinence until the final 4 weeks of the treatment period compared with those who initiated continuous abstinence by their quit date. Participants who reported >30 days of abstinence during the year prior to study entry were significantly more likely to relapse than those who reported 0 days of abstinence (OR=2.38, 95% CI: 1.17-5.04; p=.013).
Conclusion: Results of these analyses suggest that the ability to quit smoking on the initial quit date and maintain abstinence throughout the treatment period is a good prognostic indicator for long-term abstinence. The relationship between post-treatment relapse and longer pretreatment periods of abstinence is counterintuitive, yet not without precedence in the literature.
Copyright (c) 2009 Elsevier Ireland Ltd. All rights reserved.
Conflict of interest statement
Figures
Similar articles
-
A discrete-event simulation of smoking-cessation strategies based on varenicline pivotal trial data.Pharmacoeconomics. 2011 Jun;29(6):497-510. doi: 10.2165/11589230-000000000-00000. Pharmacoeconomics. 2011. PMID: 21452908
-
Efficacy of varenicline combined with nicotine replacement therapy vs varenicline alone for smoking cessation: a randomized clinical trial.JAMA. 2014 Jul;312(2):155-61. doi: 10.1001/jama.2014.7195. JAMA. 2014. PMID: 25005652 Clinical Trial.
-
Varenicline: a first-line treatment option for smoking cessation.Clin Ther. 2009 Mar;31(3):463-91. doi: 10.1016/j.clinthera.2009.03.021. Clin Ther. 2009. PMID: 19393839 Review.
-
Effect of maintenance therapy with varenicline on smoking cessation: a randomized controlled trial.JAMA. 2006 Jul 5;296(1):64-71. doi: 10.1001/jama.296.1.64. JAMA. 2006. PMID: 16820548 Clinical Trial.
-
Varenicline: a novel pharmacotherapy for smoking cessation.Drugs. 2009 Jul 9;69(10):1319-38. doi: 10.2165/00003495-200969100-00003. Drugs. 2009. PMID: 19583451 Review.
Cited by
-
Predictors of tobacco abstinence in outpatient smokers with schizophrenia or bipolar disorder treated with varenicline and cognitive behavioral smoking cessation therapy.Addict Behav. 2017 Aug;71:89-95. doi: 10.1016/j.addbeh.2017.02.028. Epub 2017 Feb 24. Addict Behav. 2017. PMID: 28285208 Free PMC article.
-
Do tobacco regulatory and economic factors influence smoking cessation outcomes? A post-hoc analysis of the multinational EAGLES randomised controlled trial.BMJ Open. 2024 Sep 20;14(9):e079092. doi: 10.1136/bmjopen-2023-079092. BMJ Open. 2024. PMID: 39306350 Free PMC article. Clinical Trial.
-
Immediate versus delayed quitting and rates of relapse among smokers treated successfully with varenicline, bupropion SR or placebo.Addiction. 2010 Nov;105(11):2002-13. doi: 10.1111/j.1360-0443.2010.03058.x. Epub 2010 Sep 1. Addiction. 2010. PMID: 20819082 Free PMC article. Clinical Trial.
-
Nicotine receptor partial agonists for smoking cessation.Cochrane Database Syst Rev. 2016 May 9;2016(5):CD006103. doi: 10.1002/14651858.CD006103.pub7. Cochrane Database Syst Rev. 2016. Update in: Cochrane Database Syst Rev. 2023 May 5;5:CD006103. doi: 10.1002/14651858.CD006103.pub8. PMID: 27158893 Free PMC article. Updated.
-
What Factors Influence Non-Adherence to the Smoking Cessation Program?Turk Thorac J. 2019 Apr 1;20(3):168-174. doi: 10.5152/TurkThoracJ.2018.18040. Print 2019 Jul. Turk Thorac J. 2019. PMID: 30986171 Free PMC article.
References
-
- Bandura A. Self-efficacy: Toward a unifying theory of behavioral change. Psychol Rev. 1977;84:191–215. - PubMed
-
- Brandon TH, Tiffany ST, Obremski KM, Baker TB. Postcessation cigarette use: The process of relapse. Addict Behav. 1990;15:105–114. - PubMed
-
- Carlson LE, Taenzer P, Koopmans J, Bultz BD. Eight-year follow-up of a community-based large group behavioral smoking cessation intervention. Addict Behav. 2000;25:725–741. - PubMed
-
- Centers for Disease Control and Prevention. Cigarette smoking among adults—United States. MMWR. Morbidity and Mortality Weekly Report. 2000;51:642–645. - PubMed
-
- Cui Y, Wen W, Moriarty CJ, Levine RS. Risk factors and their effects on the dynamic process of smoking relapse among veteran smokers. Behav Res Ther. 2006;44:967–981. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical