Constitutive genetic deletion of the growth regulator Nogo-A induces schizophrenia-related endophenotypes
- PMID: 20071518
- PMCID: PMC6633005
- DOI: 10.1523/JNEUROSCI.4393-09.2010
Constitutive genetic deletion of the growth regulator Nogo-A induces schizophrenia-related endophenotypes
Abstract
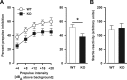
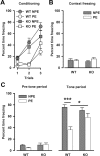
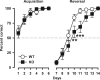
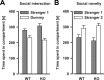
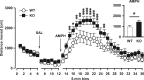
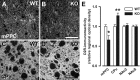
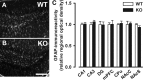
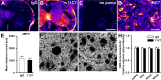
The membrane protein Nogo-A, which is predominantly expressed by oligodendrocytes in the adult CNS and by neurons mainly during development, is well known for limiting neurite outgrowth and regeneration in the injured mammalian CNS. In addition, it has recently been proposed that abnormal Nogo-A expression or Nogo receptor (NgR) mutations may confer genetic risks for neuropsychiatric disorders of presumed neurodevelopmental origin, such as schizophrenia. We therefore evaluated whether Nogo-A deletion may lead to schizophrenia-like abnormalities in a mouse model of genetic Nogo-A deficiency. Here, we show that systemic, lifelong knock-out of the Nogo-A gene can lead to specific behavioral abnormalities resembling schizophrenia-related endophenotypes: deficient sensorimotor gating, disrupted latent inhibition, perseverative behavior, and increased sensitivity to the locomotor stimulating effects of amphetamine. These behavioral phenotypes were accompanied by altered monoaminergic transmitter levels in specific striatal and limbic structures, as well as changes in dopamine D2 receptor expression in the same brain regions. Nogo-A deletion was further associated with elevated expression of growth-related markers. In contrast, acute antibody-mediated Nogo-A neutralization in adult wild-type mice failed to produce such phenotypes, suggesting that the phenotypes observed in the knock-out mice might be of developmental origin, and that Nogo-A normally subserves critical functions in neurodevelopment. This study provides the first experimental demonstration that Nogo-A bears neuropsychiatric relevance, and alterations in its expression may be one etiological factor in schizophrenia and related disorders.
Figures

Similar articles
-
Behavioral characterization of mice lacking the neurite outgrowth inhibitor Nogo-A.Genes Brain Behav. 2009 Mar;8(2):181-92. doi: 10.1111/j.1601-183X.2008.00460.x. Epub 2008 Dec 3. Genes Brain Behav. 2009. PMID: 19077178
-
Deletion of CB2 cannabinoid receptor induces schizophrenia-related behaviors in mice.Neuropsychopharmacology. 2011 Jun;36(7):1489-504. doi: 10.1038/npp.2011.34. Epub 2011 Mar 23. Neuropsychopharmacology. 2011. PMID: 21430651 Free PMC article.
-
Schizophrenia-like features in transgenic mice overexpressing human HO-1 in the astrocytic compartment.J Neurosci. 2012 Aug 8;32(32):10841-53. doi: 10.1523/JNEUROSCI.6469-11.2012. J Neurosci. 2012. PMID: 22875919 Free PMC article.
-
Effects of SPAK knockout on sensorimotor gating, novelty exploration, and brain area-dependent expressions of NKCC1 and KCC2 in a mouse model of schizophrenia.Prog Neuropsychopharmacol Biol Psychiatry. 2015 Aug 3;61:30-6. doi: 10.1016/j.pnpbp.2015.03.007. Epub 2015 Mar 19. Prog Neuropsychopharmacol Biol Psychiatry. 2015. PMID: 25797415 Review.
-
Nogo and Nogo receptor: relevance to schizophrenia?Neurobiol Dis. 2013 Jun;54:150-7. doi: 10.1016/j.nbd.2013.01.011. Epub 2013 Jan 28. Neurobiol Dis. 2013. PMID: 23369871 Review.
Cited by
-
Nogo-A and LINGO-1: Two Important Targets for Remyelination and Regeneration.Int J Mol Sci. 2023 Feb 24;24(5):4479. doi: 10.3390/ijms24054479. Int J Mol Sci. 2023. PMID: 36901909 Free PMC article. Review.
-
N-Methyl-d-Aspartate Receptor - Nitric Oxide Synthase Pathway in the Cortex of Nogo-A-Deficient Rats in Relation to Brain Laterality and Schizophrenia.Front Behav Neurosci. 2013 Aug 12;7:90. doi: 10.3389/fnbeh.2013.00090. eCollection 2013. Front Behav Neurosci. 2013. PMID: 23964213 Free PMC article.
-
Loss of Nogo-A, encoded by the schizophrenia risk gene Rtn4, reduces mGlu3 expression and causes hyperexcitability in hippocampal CA3 circuits.PLoS One. 2018 Jul 24;13(7):e0200896. doi: 10.1371/journal.pone.0200896. eCollection 2018. PLoS One. 2018. PMID: 30040841 Free PMC article.
-
Adenosine hypothesis of schizophrenia--opportunities for pharmacotherapy.Neuropharmacology. 2012 Mar;62(3):1527-43. doi: 10.1016/j.neuropharm.2011.01.048. Epub 2011 Feb 17. Neuropharmacology. 2012. PMID: 21315743 Free PMC article. Review.
-
Nogo limits neural plasticity and recovery from injury.Curr Opin Neurobiol. 2014 Aug;27:53-60. doi: 10.1016/j.conb.2014.02.011. Epub 2014 Mar 12. Curr Opin Neurobiol. 2014. PMID: 24632308 Free PMC article. Review.
References
-
- Abi-Dargham A. Alterations of serotonin transmission in schizophrenia. Int Rev Neurobiol. 2007;78:133–164. - PubMed
-
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders (DSM-IV) Washington, DC: APA; 2000.
-
- Arguello PA, Gogos JA. Modeling madness in mice: one piece at a time. Neuron. 2006;52:179–196. - PubMed
-
- Baruch I, Hemsley DR, Gray JA. Differential performance of acute and chronic schizophrenics in a latent inhibition task. J Nerv Ment Dis. 1988;176:598–606. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
