Conduction block in PMP22 deficiency
- PMID: 20071523
- PMCID: PMC3676309
- DOI: 10.1523/JNEUROSCI.4264-09.2010
Conduction block in PMP22 deficiency
Abstract
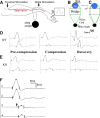
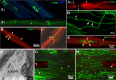
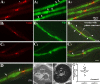
Patients with PMP22 deficiency present with focal sensory and motor deficits when peripheral nerves are stressed by mechanical force. It has been hypothesized that these focal deficits are due to mechanically induced conduction block (CB). To test this hypothesis, we induced 60-70% CB (defined by electrophysiological criteria) by nerve compression in an authentic mouse model of hereditary neuropathy with liability to pressure palsies (HNPP) with an inactivation of one of the two pmp22 alleles (pmp22(+/-)). Induction time for the CB was significantly shorter in pmp22(+/-) mice than that in pmp22(+/+) mice. This shortened induction was also found in myelin-associated glycoprotein knock-out mice, but not in the mice with deficiency of myelin protein zero, a major structural protein of compact myelin. Pmp22(+/-) nerves showed intact tomacula with no segmental demyelination in both noncompressed and compressed conditions, normal molecular architecture, and normal concentration of voltage-gated sodium channels by [(3)H]-saxitoxin binding assay. However, focal constrictions were observed in the axonal segments enclosed by tomacula, a pathological hallmark of HNPP. The constricted axons increase axial resistance to action potential propagation, which may hasten the induction of CB in Pmp22 deficiency. Together, these results demonstrate that a function of Pmp22 is to protect the nerve from mechanical injury.
Figures

Similar articles
-
Molecular regulators of nerve conduction - Lessons from inherited neuropathies and rodent genetic models.Exp Neurol. 2015 May;267:209-18. doi: 10.1016/j.expneurol.2015.03.009. Epub 2015 Mar 17. Exp Neurol. 2015. PMID: 25792482 Free PMC article. Review.
-
Abnormal junctions and permeability of myelin in PMP22-deficient nerves.Ann Neurol. 2014 Feb;75(2):255-65. doi: 10.1002/ana.24086. Epub 2014 Feb 20. Ann Neurol. 2014. PMID: 24339129 Free PMC article.
-
Hypermyelination and demyelinating peripheral neuropathy in Pmp22-deficient mice.Nat Genet. 1995 Nov;11(3):274-80. doi: 10.1038/ng1195-274. Nat Genet. 1995. PMID: 7581450
-
Heterozygous peripheral myelin protein 22-deficient mice are affected by a progressive demyelinating tomaculous neuropathy.J Neurosci. 1997 Jun 15;17(12):4662-71. doi: 10.1523/JNEUROSCI.17-12-04662.1997. J Neurosci. 1997. PMID: 9169527 Free PMC article.
-
Charcot-Marie-Tooth disease and related inherited neuropathies.Medicine (Baltimore). 1996 Sep;75(5):233-50. doi: 10.1097/00005792-199609000-00001. Medicine (Baltimore). 1996. PMID: 8862346 Review.
Cited by
-
Electrophysiological comparison between males and females in HNPP.Neurol Sci. 2013 Aug;34(8):1429-32. doi: 10.1007/s10072-012-1258-8. Epub 2012 Dec 4. Neurol Sci. 2013. PMID: 23207550
-
Evaluation of dermal myelinated nerve fibers in diabetes mellitus.J Peripher Nerv Syst. 2013 Jun;18(2):162-7. doi: 10.1111/jns5.12027. J Peripher Nerv Syst. 2013. PMID: 23781963 Free PMC article.
-
Nonrecurrent PMP22-RAI1 contiguous gene deletions arise from replication-based mechanisms and result in Smith-Magenis syndrome with evident peripheral neuropathy.Hum Genet. 2016 Oct;135(10):1161-74. doi: 10.1007/s00439-016-1703-5. Epub 2016 Jul 7. Hum Genet. 2016. PMID: 27386852 Free PMC article.
-
Molecular regulators of nerve conduction - Lessons from inherited neuropathies and rodent genetic models.Exp Neurol. 2015 May;267:209-18. doi: 10.1016/j.expneurol.2015.03.009. Epub 2015 Mar 17. Exp Neurol. 2015. PMID: 25792482 Free PMC article. Review.
-
Functional Recovery Occurs Even After Partial Remyelination of Axon-Meshed Median and Ulnar Nerves in Mice.Neurochem Res. 2019 Sep;44(9):2230-2236. doi: 10.1007/s11064-019-02863-9. Epub 2019 Aug 26. Neurochem Res. 2019. PMID: 31486011
References
-
- Adlkofer K, Martini R, Aguzzi A, Zielasek J, Toyka KV, Suter U. Hypermyelination and demyelinating peripheral neuropathy in Pmp22-deficient mice. Nat Genet. 1995;11:274–280. - PubMed
-
- Amici SA, Dunn WA, Jr, Notterpek L. Developmental abnormalities in the nerves of peripheral myelin protein 22-deficient mice. J Neurosci Res. 2007;85:238–249. - PubMed
-
- Bai Y, Ianokova E, Pu Q, Ghandour K, Levinson R, Martin JJ, Ceuterick-de Groote C, Mazanec R, Seeman P, Shy ME, Li J. R69C mutation in P0 gene alters myelination and ion channel subtypes. Arch Neurol. 2006;63:1787–1794. - PubMed
-
- Bishop DL, Misgeld T, Walsh MK, Gan WB, Lichtman JW. Axon branch removal at developing synapses by axosome shedding. Neuron. 2004;44:651–661. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
