Human cytomegalovirus infection causes premature and abnormal differentiation of human neural progenitor cells
- PMID: 20071566
- PMCID: PMC2838134
- DOI: 10.1128/JVI.02161-09
Human cytomegalovirus infection causes premature and abnormal differentiation of human neural progenitor cells
Abstract
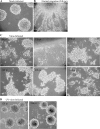
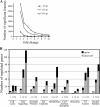
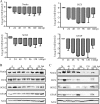
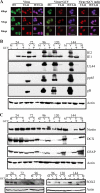
Congenital human cytomegalovirus (HCMV) infection is a leading cause of birth defects, largely manifested as central nervous system (CNS) disorders. The principal site of manifestations in the mouse model is the fetal brain's neural progenitor cell (NPC)-rich subventricular zone. Our previous human NPC studies found these cells to be fully permissive for HCMV and a useful in vitro model system. In continuing work, we observed that under culture conditions favoring maintenance of multipotency, infection caused NPCs to quickly and abnormally differentiate. This phenotypic change required active viral transcription. Whole-genome expression analysis found rapid downregulation of genes that maintain multipotency and establish NPCs' neural identity. Quantitative PCR, Western blot, and immunofluorescence assays confirmed that the mRNA and protein levels of four hallmark NPC proteins (nestin, doublecortin, sex-determining homeobox 2, and glial fibrillary acidic protein) were decreased by HCMV infection. The decreases required active viral replication and were due, at least in part, to proteasomal degradation. Our results suggest that HCMV infection causes in utero CNS defects by inducing both premature and abnormal differentiation of NPCs.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

