An empty E1, E3, E4 adenovirus vector protects photoreceptors from light-induced degeneration
- PMID: 20072633
- PMCID: PMC2802415
- DOI: 10.1007/s12177-008-9004-4
An empty E1, E3, E4 adenovirus vector protects photoreceptors from light-induced degeneration
Abstract
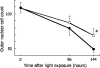
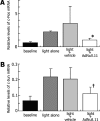
We have previously identified a neuroprotective effect associated with empty (E1(-), E3(-), E4(-)) adenovirus vector delivery in a model of light-induced, photoreceptor cell death. In this study, we further characterize this protective effect in light-injured retina and investigate its molecular basis. Dark-adapted BALB/c mice, aged 6-8 weeks, were exposed to standardized, intense fluorescent light for 96 or 144 h. Prior to dark adaptation, all mice received intravitreous injection of 1 x 10(9) particles of an empty (E1(-), E3(-), E4(-)) adenovirus vector in one eye and vehicle in the other. Following light challenge of 96 or 144 h, histopathological analysis and quantitative photoreceptor cell counts were conducted. Semiquantitative assessment of messenger ribonucleic acid (mRNA) for the apoptosis related genes: p50, p65, IkBa, caspase-1, caspase-3, Bad, c-Jun, Bax, Bak, Bcl-2, c-Fos, and p53 using quantitative reverse transcriptase polymerase chain reaction was performed on eyes following 12 h of light exposure. Following 96 h of light exposure, the photoreceptor cell density for E1(-), E3(-), E4(-) adenovirus vector and vehicle-injected eyes were 87.5 +/- 9.5 and 79.3 +/- 10.1, respectively, (p = 0.79). After 144 h of light exposure, the photoreceptor cell density was preserved in vector-injected eyes as compared to vehicle treated eyes, 68.9 +/- 10.0 and 49.2 +/- 4.6, respectively (p = 0.016). Relative mRNA levels of c-Fos and c-Jun at 12-h light exposure after injection differed significantly between vector- and vehicle-injected eyes (p = 0.036, 0.016, respectively). The expression of the other apoptosis-related genes evaluated was not significantly affected. This study investigates the molecular basis of photoreceptor neuroprotective pathway induction associated with E1(-), E3(-), E4(-) adenovirus vectors. The results indicate that empty adenovirus vectors protect photoreceptors from light-induced degeneration by the modulation of apoptotic pathways. Gene expression changes suggest that the suppression of c-Fos and c-Jun upregulation contributes significantly to the neuroprotective effect. Understanding the molecular basis of the neuroprotective pathway induction in photoreceptors is critical to the development of novel therapies for retinal degenerations.
Keywords: Adenoviral vector; Apoptosis; Neuroprotection; Null effect; Retinal light damage.
Figures

Similar articles
-
Intraocular gene transfer of pigment epithelium-derived factor rescues photoreceptors from light-induced cell death.J Cell Physiol. 2005 Feb;202(2):570-8. doi: 10.1002/jcp.20155. J Cell Physiol. 2005. PMID: 15316929
-
Reduced toxicity, attenuated immunogenicity and efficient mediation of human p53 gene expression in vivo by an adenovirus vector with deleted E1-E3 and inactivated E4 by GAL4-TATA promoter replacement.Gene Ther. 1999 Mar;6(3):393-402. doi: 10.1038/sj.gt.3300825. Gene Ther. 1999. PMID: 10435089
-
Gene expression in the mouse retina: the effect of damaging light.Mol Vis. 2000 Dec 13;6:252-60. Mol Vis. 2000. PMID: 11134582
-
Generation and characterization of E1/E2a/E3/E4-deficient adenoviral vectors encoding human factor VIII.Mol Ther. 2001 Mar;3(3):329-36. doi: 10.1006/mthe.2001.0264. Mol Ther. 2001. PMID: 11273775
-
Molecular mechanisms of light-induced photoreceptor apoptosis and neuroprotection for retinal degeneration.Prog Retin Eye Res. 2005 Mar;24(2):275-306. doi: 10.1016/j.preteyeres.2004.08.002. Epub 2004 Nov 11. Prog Retin Eye Res. 2005. PMID: 15610977 Review.
References
-
- Hauswirth WW, Beaufrere L. Ocular gene therapy: quo vadis? Invest Ophthalmol Vis Sci. 2000;41:2821–6. - PubMed
-
- Reichel MB, Bainbridge J, Baker D, Thrasher AJ, Bhattacharya SS, Ali RR. An immune response after intraocular administration of an adenoviral vector containing a beta galactosidase reporter gene slows retinal degeneration in the rd mouse. Br J Ophthalmol. 2001;85:341–4. doi: 10.1136/bjo.85.3.341. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
