A process for microbial hydrocarbon synthesis: Overproduction of fatty acids in Escherichia coli and catalytic conversion to alkanes
- PMID: 20073090
- PMCID: PMC3833807
- DOI: 10.1002/bit.22660
A process for microbial hydrocarbon synthesis: Overproduction of fatty acids in Escherichia coli and catalytic conversion to alkanes
Abstract
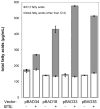
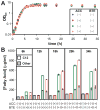
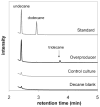
The development of renewable alternatives to diesel and jet fuels is highly desirable for the heavy transportation sector, and would offer benefits over the production and use of short-chain alcohols for personal transportation. Here, we report the development of a metabolically engineered strain of Escherichia coli that overproduces medium-chain length fatty acids via three basic modifications: elimination of beta-oxidation, overexpression of the four subunits of acetyl-CoA carboxylase, and expression of a plant acyl-acyl carrier protein (ACP) thioesterase from Umbellularia californica (BTE). The expression level of BTE was optimized by comparing fatty acid production from strains harboring BTE on plasmids with four different copy numbers. Expression of BTE from low copy number plasmids resulted in the highest fatty acid production. Up to a seven-fold increase in total fatty acid production was observed in engineered strains over a negative control strain (lacking beta-oxidation), with a composition dominated by C(12) and C(14) saturated and unsaturated fatty acids. Next, a strategy for producing undecane via a combination of biotechnology and heterogeneous catalysis is demonstrated. Fatty acids were extracted from a culture of an overproducing strain into an alkane phase and fed to a Pd/C plug flow reactor, where the extracted fatty acids were decarboxylated into saturated alkanes. The result is an enriched alkane stream that can be recycled for continuous extractions. Complete conversion of C(12) fatty acids extracted from culture to alkanes has been demonstrated yielding a concentration of 0.44 g L(-1) (culture volume) undecane.
Figures

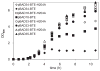
References
-
- Antoine R, Locht C. Isolation and molecular characterization of a novel broad-host-range plasmid from Bordetella bronchiseptica with sequence similarities to plasmids from Gram-positive organisms. Mol Microbiol. 1992;6:1785–1799. - PubMed
-
- Atsumi S, Hanai T, Liao JC. Non-fermentative pathways for synthesis of branched-chain higher alcohols as biofuels. Nature. 2008;451:86–89. - PubMed
-
- Banerjee A, Sharma R, Chisti Y, Banerjee U. Botryococcus braunii: A renewable source of hydrocarbons and other chemicals. Crit Rev Microbiol. 2002;22:245–279. - PubMed
-
- Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37:911–917. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

