Cell aggregation induces phosphorylation of PECAM-1 and Pyk2 and promotes tumor cell anchorage-independent growth
- PMID: 20074345
- PMCID: PMC2820017
- DOI: 10.1186/1476-4598-9-7
Cell aggregation induces phosphorylation of PECAM-1 and Pyk2 and promotes tumor cell anchorage-independent growth
Abstract
Background: Apoptosis caused by inadequate or inappropriate cell-matrix interactions is defined as anoikis. Although transformed cells are known to be anoikis-resistant, the underlying mechanisms have not been well understood. We investigated the mechanisms of anoikis resistance of tumor cells.
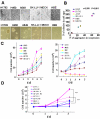
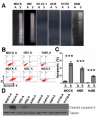
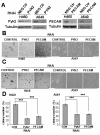
Results: We observed that cell aggregation in suspension promoted cell survival and proliferation. We demonstrated a correlation between tumor cell aggregation in suspension and cell growth in soft agar. Analysis of tyrosine kinase-mediated cell survival and growth signaling pathways revealed increased levels of tyrosine-phosphorylation of PECAM-1 and Pyk2 in cell aggregates. We also showed that PECAM-1 and Pyk2 physically interact with each other, and that PECAM-1 carrying a deletion of exons 11-16 could no longer bind to Pyk2. Furthermore, RNA interference-mediated reduction of Pyk2 and PECAM-1 protein levels reduced cell aggregation and inhibited the growth of tumor cells in soft agar.
Conclusions: The data demonstrated that Pyk2 and PECAM-1 were critical mediators of both anchorage-independent growth and anoikis resistance in tumor cells.
Figures


Similar articles
-
Wheat germ agglutinin-induced platelet activation via platelet endothelial cell adhesion molecule-1: involvement of rapid phospholipase C gamma 2 activation by Src family kinases.Biochemistry. 2001 Oct 30;40(43):12992-3001. doi: 10.1021/bi0109459. Biochemistry. 2001. PMID: 11669637
-
Glucocorticoids induce osteocyte apoptosis by blocking focal adhesion kinase-mediated survival. Evidence for inside-out signaling leading to anoikis.J Biol Chem. 2007 Aug 17;282(33):24120-30. doi: 10.1074/jbc.M611435200. Epub 2007 Jun 20. J Biol Chem. 2007. PMID: 17581824
-
Squamous cell carcinoma cell aggregates escape suspension-induced, p53-mediated anoikis: fibronectin and integrin alphav mediate survival signals through focal adhesion kinase.J Biol Chem. 2004 Nov 12;279(46):48342-9. doi: 10.1074/jbc.M407953200. Epub 2004 Aug 24. J Biol Chem. 2004. PMID: 15331608
-
Role of Pyk2 in Human Cancers.Med Sci Monit. 2018 Nov 14;24:8172-8182. doi: 10.12659/MSM.913479. Med Sci Monit. 2018. PMID: 30425234 Free PMC article. Review.
-
Mechanisms of PECAM-1-mediated cytoprotection and implications for cancer cell survival.Leuk Lymphoma. 2005 Oct;46(10):1409-21. doi: 10.1080/10428190500126091. Leuk Lymphoma. 2005. PMID: 16194886 Review.
Cited by
-
Controlling Cancer Cell Behavior by Improving the Stiffness of Gastric Tissue-Decellularized ECM Bioink With Cellulose Nanoparticles.Front Bioeng Biotechnol. 2021 Mar 17;9:605819. doi: 10.3389/fbioe.2021.605819. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 33816446 Free PMC article.
-
Deciphering anoikis resistance and identifying prognostic biomarkers in clear cell renal cell carcinoma epithelial cells.Sci Rep. 2024 May 27;14(1):12044. doi: 10.1038/s41598-024-62978-0. Sci Rep. 2024. PMID: 38802480 Free PMC article.
-
A circulating tumor cell cluster-based model for tumor metastasis (Hypothesis).Oncol Lett. 2016 Dec;12(6):4891-4895. doi: 10.3892/ol.2016.5358. Epub 2016 Nov 7. Oncol Lett. 2016. PMID: 28105198 Free PMC article.
-
The role of platelets in tumor immune evasion and metastasis: mechanisms and therapeutic implications.Cancer Cell Int. 2025 Jul 11;25(1):258. doi: 10.1186/s12935-025-03877-w. Cancer Cell Int. 2025. PMID: 40646579 Free PMC article. Review.
-
Integrin αVβ1-activated PYK2 promotes the progression of non-small-cell lung cancer via the STAT3-VGF axis.Cell Commun Signal. 2024 Jun 6;22(1):313. doi: 10.1186/s12964-024-01639-1. Cell Commun Signal. 2024. PMID: 38844957 Free PMC article.
References
-
- Steuer AF, Ting RC. Formation of larger cell aggregates by transformed cells: an in vitro index of cell transformation. J Natl Cancer Inst. 1976;56:1279–1280. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

