PIKE-A is required for prolactin-mediated STAT5a activation in mammary gland development
- PMID: 20075866
- PMCID: PMC2837170
- DOI: 10.1038/emboj.2009.406
PIKE-A is required for prolactin-mediated STAT5a activation in mammary gland development
Abstract
PI 3-kinase enhancer A (PIKE-A) is critical for the activation of Akt signalling, and has an essential function in promoting cancer cell survival. However, its physiological functions are poorly understood. Here, we show that PIKE-A directly associates with both signal transducer and activator of transcription 5a (STAT5a) and prolactin (PRL) receptor, which is essential for PRL-provoked STAT5a activation and the subsequent gene transcription. Depletion of PIKE-A in HC11 epithelial cells diminished PRL-induced STAT5 activation and cyclin D1 expression, resulting in profoundly impaired cell proliferation in vitro. To confirm the function of PIKE-A in PRL signalling in vivo, we generated PIKE knockout (PIKE-/-) mice. PIKE-/- mice displayed a severe lactation defect that was characterized by enhanced apoptosis and impaired proliferation of mammary epithelial cells. At parturition, STAT5 activation and cyclin D1 expression were substantially reduced in the mammary epithelium of PIKE-/- mice. The defective mammary gland development in PIKE-/- mice was rescued by overexpression of a mammary-specific cyclin D1 transgene. These data establish a critical function for PIKE-A in mediating PRL functions.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

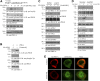
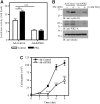
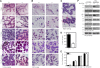
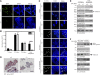


Similar articles
-
Basal activation of transcription factor signal transducer and activator of transcription (Stat5) in nonpregnant mouse and human breast epithelium.Mol Endocrinol. 2002 May;16(5):1108-24. doi: 10.1210/mend.16.5.0839. Mol Endocrinol. 2002. PMID: 11981045
-
Zinc Finger Homeodomain Factor Zfhx3 Is Essential for Mammary Lactogenic Differentiation by Maintaining Prolactin Signaling Activity.J Biol Chem. 2016 Jun 10;291(24):12809-12820. doi: 10.1074/jbc.M116.719377. Epub 2016 Apr 20. J Biol Chem. 2016. PMID: 27129249 Free PMC article.
-
CUZD1 is a critical mediator of the JAK/STAT5 signaling pathway that controls mammary gland development during pregnancy.PLoS Genet. 2017 Mar 9;13(3):e1006654. doi: 10.1371/journal.pgen.1006654. eCollection 2017 Mar. PLoS Genet. 2017. PMID: 28278176 Free PMC article.
-
Signal transducer and activator of transcription 5 as a key signaling pathway in normal mammary gland developmental biology and breast cancer.Breast Cancer Res. 2011 Oct 12;13(5):220. doi: 10.1186/bcr2921. Breast Cancer Res. 2011. PMID: 22018398 Free PMC article. Review.
-
The role of prolactin and growth hormone in mammary gland development.Mol Cell Endocrinol. 2002 Nov 29;197(1-2):127-31. doi: 10.1016/s0303-7207(02)00286-1. Mol Cell Endocrinol. 2002. PMID: 12431805 Review.
Cited by
-
Autocrine prolactin: an emerging market for homegrown (prolactin) despite the imports.Genes Dev. 2012 Oct 15;26(20):2253-8. doi: 10.1101/gad.204636.112. Genes Dev. 2012. PMID: 23070811 Free PMC article.
-
Muscle-generated BDNF is a sexually dimorphic myokine that controls metabolic flexibility.Sci Signal. 2019 Aug 13;12(594):eaau1468. doi: 10.1126/scisignal.aau1468. Sci Signal. 2019. PMID: 31409756 Free PMC article.
-
Tumor Necrosis Factor-α Promotes Phosphoinositide 3-Kinase Enhancer A and AMP-Activated Protein Kinase Interaction to Suppress Lipid Oxidation in Skeletal Muscle.Diabetes. 2017 Jul;66(7):1858-1870. doi: 10.2337/db16-0270. Epub 2017 Apr 12. Diabetes. 2017. PMID: 28404596 Free PMC article.
-
Phosphoinositide 3-kinase enhancer (PIKE) in the brain: is it simply a phosphoinositide 3-kinase/Akt enhancer?Rev Neurosci. 2012 Jan 26;23(2):153-61. doi: 10.1515/revneuro-2011-0066. Rev Neurosci. 2012. PMID: 22499674 Free PMC article. Review.
-
The roles of PIKE in tumorigenesis.Acta Pharmacol Sin. 2013 Aug;34(8):991-7. doi: 10.1038/aps.2013.71. Epub 2013 Jun 17. Acta Pharmacol Sin. 2013. PMID: 23770988 Free PMC article. Review.
References
-
- Ahn JY, Rong R, Kroll TG, Van Meir EG, Snyder SH, Ye K (2004b) PIKE (phosphatidylinositol 3-kinase enhancer)-A GTPase stimulates Akt activity and mediates cellular invasion. J Biol Chem 279: 16441–16451 - PubMed
-
- Brisken C (2002) Hormonal control of alveolar development and its implications for breast carcinogenesis. J Mammary Gland Biol Neoplasia 7: 39–48 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

