(-)-Epigallocatechin gallate sensitizes breast cancer cells to paclitaxel in a murine model of breast carcinoma
- PMID: 20078855
- PMCID: PMC2880428
- DOI: 10.1186/bcr2473
(-)-Epigallocatechin gallate sensitizes breast cancer cells to paclitaxel in a murine model of breast carcinoma
Abstract
Introduction: Paclitaxel (Taxol) is a microtubule-targeted agent that is widely used for cancer treatment. However, resistance to paclitaxel is frequently encountered in the clinic. There is increasing interest in identifying compounds that may increase the sensitivity to conventional chemotherapeutic agents. In this study, we investigated whether green tea polyphenol (-)-epigallocatechin gallate (EGCG) could sensitize breast carcinoma to paclitaxel in vivo.
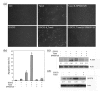
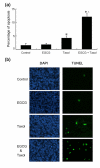
Methods: Breast cancer cells were treated with or without EGCG and paclitaxel followed by detection of cell survival and apoptosis. c-Jun NH2-terminal kinase (JNK) phosphorylation and glucose-regulated protein 78 (GRP78) expression were detected by Western blotting. For in vivo study, 4T1 breast cancer cells were inoculated into Balb/c mice to establish a transplantation model. The tumor-bearing mice were treated with or without EGCG (30 mg/kg, i.p.) and paclitaxel (10 mg/kg, i.p.). Tumor growth was monitored. Apoptosis in tumor tissues was detected. Cell lysates from tumors were subjected to Western blot analysis of GRP78 expression and JNK phosphorylation.
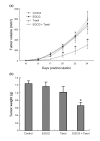
Results: EGCG synergistically sensitized breast cancer cells to paclitaxel in vitro and in vivo. EGCG in combination with paclitaxel significantly induced 4T1 cells apoptosis compared with each single treatment. When tumor-bearing mice were treated with paclitaxel in combination with EGCG, tumor growth was significantly inhibited, whereas the single-agent activity for paclitaxel or EGCG was poor. EGCG overcame paclitaxel-induced GRP78 expression and potentiated paclitaxel-induced JNK phosphorylation in 4T1 cells both in vitro and in vivo.
Conclusions: EGCG may be used as a sensitizer to enhance the cytotoxicity of paclitaxel.
Figures
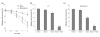


References
-
- McGuire WP, Rowinsky EK, Rosenshein NB, Grumbine FC, Ettinger DS, Armstrong DK, Donehower RC. Taxol: a unique antineoplastic agent with significant activity in advanced ovarian epithelial neoplasms. Ann Intern Med. 1989;111:273–279. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

