Identification of calcium-binding proteins associated with the human sperm plasma membrane
- PMID: 20078857
- PMCID: PMC2822784
- DOI: 10.1186/1477-7827-8-6
Identification of calcium-binding proteins associated with the human sperm plasma membrane
Abstract
Background: The precise composition of the human sperm plasma membrane, the molecular interactions that define domain specific functions, and the regulation of membrane associated proteins during the capacitation process, still remain to be fully understood. Here, we investigated the repertoire of calcium-regulated proteins associated with the human sperm plasma membrane.
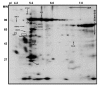
Methods: Surface specific radioiodination was combined with two-dimensional gel electrophoresis, a 45Ca-overlay assay, computer assisted image analysis and mass spectrometry to identify calcium-binding proteins exposed on the human sperm surface.
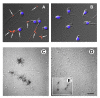
Results: Nine acidic 45Ca-binding sperm proteins were excised from stained preparative 2D gels and identified by mass spectrometry. Five of the calcium binding proteins; HSPA2 (HSP70-1), HSPA5 (Bip), HYOU1 (ORP150), serum amyloid P-component (SAP) and protein kinase C substrate 80K-H (80K-H) were found to be accessible to Iodo-Bead catalyzed 125I-labelling on the surface of intact human sperm. Agglutination and immunofluorescence analysis confirmed that SAP is situated on the plasma membrane of intact, motile sperm as well as permeabilized cells. Western blot analysis showed increased phosphorylation of human sperm 80K-H protein following in vitro capacitation. This is the first demonstration of the 80K-H protein in a mammalian sperm.
Conclusion: The presence of SAP on the surface of mature sperm implies that SAP has a physiological role in reproduction, which is thought to be in the removal of spermatozoa from the female genital tract via phagocytosis. Since 80K-H is a Ca2+-sensor recently implicated in the regulation of both inositol 1,4,5-trisphosphate receptor and transient receptor potential (TRP) cation channel activities, its detection in sperm represents the first direct signaling link between PKC and store-operated calcium channels identified in human sperm.
Figures



Similar articles
-
Heat shock proteins on the human sperm surface.J Reprod Immunol. 2010 Jan;84(1):32-40. doi: 10.1016/j.jri.2009.09.006. Epub 2009 Dec 3. J Reprod Immunol. 2010. PMID: 19962198 Free PMC article.
-
Proteomic characterization of human sperm plasma membrane-associated proteins and their role in capacitation.Andrology. 2020 Jan;8(1):171-180. doi: 10.1111/andr.12627. Epub 2019 Apr 19. Andrology. 2020. PMID: 31002753
-
The impact of oxidative stress on chaperone-mediated human sperm-egg interaction.Hum Reprod. 2015 Nov;30(11):2597-613. doi: 10.1093/humrep/dev214. Epub 2015 Sep 7. Hum Reprod. 2015. PMID: 26345691
-
Intracellular calcium regulation in sperm capacitation and acrosomal reaction.Mol Cell Endocrinol. 2002 Feb 22;187(1-2):139-44. doi: 10.1016/s0303-7207(01)00704-3. Mol Cell Endocrinol. 2002. PMID: 11988321 Review.
-
Molecular physiology and pathology of Ca2+-conducting channels in the plasma membrane of mammalian sperm.Reproduction. 2005 Mar;129(3):251-62. doi: 10.1530/rep.1.00478. Reproduction. 2005. PMID: 15749952 Review.
Cited by
-
Endoplasmic reticulum chaperones and their roles in the immunogenicity of cancer vaccines.Front Oncol. 2015 Jan 6;4:379. doi: 10.3389/fonc.2014.00379. eCollection 2014. Front Oncol. 2015. PMID: 25610811 Free PMC article. Review.
-
Calciomics: integrative studies of Ca2+-binding proteins and their interactomes in biological systems.Metallomics. 2013 Jan;5(1):29-42. doi: 10.1039/c2mt20009k. Metallomics. 2013. PMID: 23235533 Free PMC article. Review.
-
Proteomic analysis reveals dysregulated cell signaling in ejaculated spermatozoa from infertile men.Asian J Androl. 2019 Mar-Apr;21(2):121-130. doi: 10.4103/aja.aja_56_18. Asian J Androl. 2019. PMID: 30381577 Free PMC article.
-
Expression of α-subunit of α-glucosidase II in adult mouse brain regions and selected organs.J Neurosci Res. 2015 Jan;93(1):82-93. doi: 10.1002/jnr.23470. Epub 2014 Aug 18. J Neurosci Res. 2015. PMID: 25131991 Free PMC article.
-
Role of Macroautophagy in Mammalian Male Reproductive Physiology.Cells. 2023 May 5;12(9):1322. doi: 10.3390/cells12091322. Cells. 2023. PMID: 37174722 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

