CD44 regulates survival and memory development in Th1 cells
- PMID: 20079666
- PMCID: PMC2858628
- DOI: 10.1016/j.immuni.2009.10.011
CD44 regulates survival and memory development in Th1 cells
Abstract
Optimal immunity to microorganisms depends upon the regulated death of clonally expanded effector cells and the survival of a cohort of cells that become memory cells. After activation of naive T cells, CD44, a widely expressed receptor for extracellular matrix components, is upregulated. High expression of CD44 remains on memory cells and despite its wide usage as a "memory marker," its function is unknown. Here we report that CD44 was essential for the generation of memory T helper 1 (Th1) cells by promoting effector cell survival. This dependency was not found in Th2, Th17, or CD8(+) T cells despite similar expression of CD44 and the absence of splice variants in all subsets. CD44 limited Fas-mediated death in Th1 cells and its ligation engaged the phosphoinositide 3-kinase-Akt kinase signaling pathway that regulates cell survival. The difference in CD44-regulated apoptosis resistance in T cell subpopulations has important implications in a broad spectrum of diseases.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures
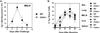




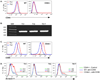

References
-
- Barnden MJ, Allison J, Heath WR, R CF. Defective TCR expression in transgenic mice constructed using cDNA based a- and b- chain genes under the control of heterologous regulatory elements. Immunol. Cell. Biol. 1998;76:34–40. - PubMed
-
- Boyman O, Purton JF, Surh CD, Sprent J. Cytokines and T-cell homeostasis. Curr. Opin. Immunol. 2007;19:320–326. - PubMed
-
- Chapman TJ, Castrucci MR, Padrick RC, Bradley LM, Topham DJ. Antigen-specific and non-specific CD4+ T cell recruitment and proliferation during influenza infection. Virology. 2005;340:296–306. - PubMed
-
- DeGrendele HC, Kosfiszer M, Estess P, Siegelman MH. CD44 activation and associated primary adhesion is inducible via T cell receptor stimulation. J. Immunol. 1997;159:2549–2553. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

