Murine leukemia virus glycosylated Gag (gPr80gag) facilitates interferon-sensitive virus release through lipid rafts
- PMID: 20080538
- PMCID: PMC2824291
- DOI: 10.1073/pnas.0908660107
Murine leukemia virus glycosylated Gag (gPr80gag) facilitates interferon-sensitive virus release through lipid rafts
Abstract
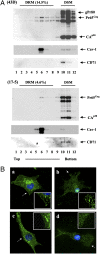
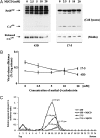
Murine leukemia viruses encode a unique form of Gag polyprotein, gPr80gag or glyco-gag. Translation of this protein is initiated from full-length viral mRNA at an upstream initiation site in the same reading frame as Pr65(gag), the precursor for internal structural (Gag) proteins. Whereas gPr80gag is evolutionarily conserved among gammaretroviruses, its mechanism of action has been unclear, although it facilitates virus production at a late assembly or release step. Here, it is shown that gPr80gag facilitates release of Moloney murine leukemia virus (M-MuLV) from cells along an IFN-sensitive pathway. In particular, gPr80gag-facilitated release occurs through lipid rafts, because gPr80gag-negative M-MuLV has a lower cholesterol content, is less sensitive to inhibition of release by the cholesterol-depleting agent MbetaCD, and there is less Pr65gag associated with detergent-resistant membranes in mutant-infected cells. gPr80gag can also facilitate the release of HIV-1-based vector particles from human 293T cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
The cellular protein La functions in enhancement of virus release through lipid rafts facilitated by murine leukemia virus glycosylated Gag.mBio. 2011 Feb 22;2(1):e00341-10. doi: 10.1128/mBio.00341-10. Print 2011. mBio. 2011. PMID: 21343359 Free PMC article.
-
Mutation in the glycosylated gag protein of murine leukemia virus results in reduced in vivo infectivity and a novel defect in viral budding or release.J Virol. 2007 Apr;81(8):3685-92. doi: 10.1128/JVI.01538-06. Epub 2007 Jan 31. J Virol. 2007. PMID: 17267509 Free PMC article.
-
Moloney murine leukemia virus glyco-gag facilitates xenotropic murine leukemia virus-related virus replication through human APOBEC3-independent mechanisms.Retrovirology. 2012 Jul 24;9:58. doi: 10.1186/1742-4690-9-58. Retrovirology. 2012. PMID: 22828015 Free PMC article.
-
Construction and characterization of Moloney murine leukemia virus mutants unable to synthesize glycosylated gag polyprotein.Proc Natl Acad Sci U S A. 1983 Oct;80(19):5965-9. doi: 10.1073/pnas.80.19.5965. Proc Natl Acad Sci U S A. 1983. PMID: 6310608 Free PMC article.
-
Structure of glycosylated and unglycosylated gag and gag-pol precursor proteins of Moloney murine leukemia virus.J Virol. 1983 Jun;46(3):841-59. doi: 10.1128/JVI.46.3.841-859.1983. J Virol. 1983. PMID: 6602220 Free PMC article.
Cited by
-
N-linked glycosylation protects gammaretroviruses against deamination by APOBEC3 proteins.J Virol. 2015 Feb;89(4):2342-57. doi: 10.1128/JVI.03330-14. Epub 2014 Dec 10. J Virol. 2015. PMID: 25505062 Free PMC article.
-
Evidence for Involvement of ADP-Ribosylation Factor 6 in Intracellular Trafficking and Release of Murine Leukemia Virus Gag.Cells. 2024 Jan 31;13(3):270. doi: 10.3390/cells13030270. Cells. 2024. PMID: 38334661 Free PMC article.
-
Murine leukemia virus glycosylated Gag blocks apolipoprotein B editing complex 3 and cytosolic sensor access to the reverse transcription complex.Proc Natl Acad Sci U S A. 2013 May 28;110(22):9078-83. doi: 10.1073/pnas.1217399110. Epub 2013 May 13. Proc Natl Acad Sci U S A. 2013. PMID: 23671100 Free PMC article.
-
Non-canonical translation in RNA viruses.J Gen Virol. 2012 Jul;93(Pt 7):1385-1409. doi: 10.1099/vir.0.042499-0. Epub 2012 Apr 25. J Gen Virol. 2012. PMID: 22535777 Free PMC article. Review.
-
The cellular protein La functions in enhancement of virus release through lipid rafts facilitated by murine leukemia virus glycosylated Gag.mBio. 2011 Feb 22;2(1):e00341-10. doi: 10.1128/mBio.00341-10. Print 2011. mBio. 2011. PMID: 21343359 Free PMC article.
References
-
- Prats AC, De Billy G, Wang P, Darlix JL. CUG initiation codon used for the synthesis of a cell surface antigen coded by the murine leukemia virus. J Mol Biol. 1989;205:363–372. - PubMed

