BBS6, BBS10, and BBS12 form a complex with CCT/TRiC family chaperonins and mediate BBSome assembly
- PMID: 20080638
- PMCID: PMC2824390
- DOI: 10.1073/pnas.0910268107
BBS6, BBS10, and BBS12 form a complex with CCT/TRiC family chaperonins and mediate BBSome assembly
Abstract
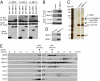
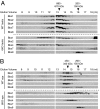
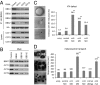
Bardet-Biedl syndrome (BBS) is a human genetic disorder resulting in obesity, retinal degeneration, polydactyly, and nephropathy. Recent studies indicate that trafficking defects to the ciliary membrane are involved in this syndrome. Here, we show that a novel complex composed of three chaperonin-like BBS proteins (BBS6, BBS10, and BBS12) and CCT/TRiC family chaperonins mediates BBSome assembly, which transports vesicles to the cilia. Chaperonin-like BBS proteins interact with a subset of BBSome subunits and promote their association with CCT chaperonins. CCT activity is essential for BBSome assembly, and knockdown of CCT chaperonins in zebrafish results in BBS phenotypes. Many disease-causing mutations found in BBS6, BBS10, and BBS12 disrupt interactions among these BBS proteins. Our data demonstrate that BBS6, BBS10, and BBS12 are necessary for BBSome assembly, and that impaired BBSome assembly contributes to the etiology of BBS phenotypes associated with the loss of function of these three BBS genes.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Intrinsic protein-protein interaction-mediated and chaperonin-assisted sequential assembly of stable bardet-biedl syndrome protein complex, the BBSome.J Biol Chem. 2012 Jun 8;287(24):20625-35. doi: 10.1074/jbc.M112.341487. Epub 2012 Apr 12. J Biol Chem. 2012. PMID: 22500027 Free PMC article.
-
Bardet-Biedl syndrome: The pleiotropic role of the chaperonin-like BBS6, 10, and 12 proteins.Am J Med Genet C Semin Med Genet. 2022 Mar;190(1):9-19. doi: 10.1002/ajmg.c.31970. Epub 2022 Apr 4. Am J Med Genet C Semin Med Genet. 2022. PMID: 35373910 Free PMC article. Review.
-
Bardet-Biedl Syndrome as a Chaperonopathy: Dissecting the Major Role of Chaperonin-Like BBS Proteins (BBS6-BBS10-BBS12).Front Mol Biosci. 2017 Jul 31;4:55. doi: 10.3389/fmolb.2017.00055. eCollection 2017. Front Mol Biosci. 2017. PMID: 28824921 Free PMC article. Review.
-
Mutations in chaperonin-like BBS genes are a major contributor to disease development in a multiethnic Bardet-Biedl syndrome patient population.J Med Genet. 2010 Jul;47(7):453-63. doi: 10.1136/jmg.2009.073205. Epub 2010 May 14. J Med Genet. 2010. PMID: 20472660
-
Identification of a novel BBS gene (BBS12) highlights the major role of a vertebrate-specific branch of chaperonin-related proteins in Bardet-Biedl syndrome.Am J Hum Genet. 2007 Jan;80(1):1-11. doi: 10.1086/510256. Epub 2006 Nov 15. Am J Hum Genet. 2007. PMID: 17160889 Free PMC article.
Cited by
-
Nephrocystin proteins NPHP5 and Cep290 regulate BBSome integrity, ciliary trafficking and cargo delivery.Hum Mol Genet. 2015 Apr 15;24(8):2185-200. doi: 10.1093/hmg/ddu738. Epub 2014 Dec 30. Hum Mol Genet. 2015. PMID: 25552655 Free PMC article.
-
Paramecium BBS genes are key to presence of channels in Cilia.Cilia. 2012 Sep 3;1(1):16. doi: 10.1186/2046-2530-1-16. Cilia. 2012. PMID: 23351336 Free PMC article.
-
Characterization of CCDC28B reveals its role in ciliogenesis and provides insight to understand its modifier effect on Bardet-Biedl syndrome.Hum Genet. 2013 Jan;132(1):91-105. doi: 10.1007/s00439-012-1228-5. Epub 2012 Sep 27. Hum Genet. 2013. PMID: 23015189
-
Peripheral blood mononuclear cell proteome changes in patients with myelodysplastic syndrome.Biomed Res Int. 2015;2015:872983. doi: 10.1155/2015/872983. Epub 2015 Apr 16. Biomed Res Int. 2015. PMID: 25969835 Free PMC article. Clinical Trial.
-
Subretinal gene therapy of mice with Bardet-Biedl syndrome type 1.Invest Ophthalmol Vis Sci. 2013 Sep 11;54(9):6118-32. doi: 10.1167/iovs.13-11673. Invest Ophthalmol Vis Sci. 2013. PMID: 23900607 Free PMC article.
References
-
- Singla V, Reiter JF. The primary cilium as the cell’s antenna: Signaling at a sensory organelle. Science. 2006;313:629–633. - PubMed
-
- Marshall WF, Nonaka S. Cilia: Tuning in to the cell's antenna. Curr Biol. 2006;16:R604–R614. - PubMed
-
- Badano JL, Mitsuma N, Beales PL, Katsanis N. The ciliopathies: An emerging class of human genetic disorders. Annu Rev Genomics Hum Genet. 2006;7:125–148. - PubMed
-
- Bisgrove BW, Yost HJ. The roles of cilia in developmental disorders and disease. Development. 2006;133:4131–4143. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

