Axl is an essential epithelial-to-mesenchymal transition-induced regulator of breast cancer metastasis and patient survival
- PMID: 20080645
- PMCID: PMC2824310
- DOI: 10.1073/pnas.0909333107
Axl is an essential epithelial-to-mesenchymal transition-induced regulator of breast cancer metastasis and patient survival
Abstract
Metastasis underlies the majority of cancer-related deaths. Thus, furthering our understanding of the molecular mechanisms that enable tumor cell dissemination is a vital health issue. Epithelial-to-mesenchymal transitions (EMTs) endow carcinoma cells with enhanced migratory and survival attributes that facilitate malignant progression. Characterization of EMT effectors is likely to yield new insights into metastasis and novel avenues for treatment. We show that the presence of the receptor tyrosine kinase Axl in primary breast cancers independently predicts strongly reduced overall patient survival, and that matched patient metastatic lesions show enhanced Axl expression. We demonstrate that Axl is strongly induced by EMT in immortalized mammary epithelial cells that establishes an autocrine signaling loop with its ligand, Gas6. Epiallelic RNA interference analysis in metastatic breast cancer cells delineated a distinct threshold of Axl expression for mesenchymal-like in vitro cell invasiveness and formation of tumors in foreign and tissue-engineered microenvironments in vivo. Importantly, in two different optical imaging-based experimental breast cancer models, Axl knockdown completely prevented the spread of highly metastatic breast carcinoma cells from the mammary gland to lymph nodes and several major organs and increased overall survival. These findings suggest that Axl represents a downstream effector of the tumor cell EMT that is required for breast cancer metastasis. Thus, the detection and targeted treatment of Axl-expressing tumors represents an important new therapeutic strategy for breast cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

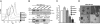
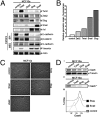
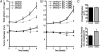
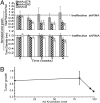

References
-
- Gupta PB, Mani S, Yang J, Hartwell K, Weinberg RA. The evolving portrait of cancer metastasis. Cold Spring Harb Symp Quant Biol. 2005;70:291–297. - PubMed
-
- Kaplan RN, Psaila B, Lyden D. Bone marrow cells in the “pre-metastatic niche”: Within bone and beyond. Cancer Metastasis Rev. 2006;25:521–529. - PubMed
-
- Thiery JP. Epithelial–mesenchymal transitions in tumour progression. Nat Rev Cancer. 2002;2:442–454. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

