Glutamate co-release at GABA/glycinergic synapses is crucial for the refinement of an inhibitory map
- PMID: 20081852
- PMCID: PMC2832847
- DOI: 10.1038/nn.2478
Glutamate co-release at GABA/glycinergic synapses is crucial for the refinement of an inhibitory map
Abstract
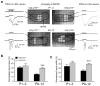
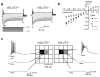
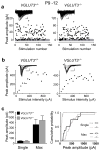
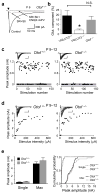
Many nonglutamatergic synaptic terminals in the mammalian brain contain the vesicular glutamate transporter 3 (VGLUT3), indicating that they co-release the excitatory neurotransmitter glutamate. However, the functional role of glutamate co-transmission at these synapses is poorly understood. In the auditory system, VGLUT3 expression and glutamate co-transmission are prominent in a developing GABA/glycinergic sound-localization pathway. We found that mice with a genetic deletion of Vglut3 (also known as Slc17a8) had disrupted glutamate co-transmission and severe impairment in the refinement of this inhibitory pathway. Specifically, loss of glutamate co-transmission disrupted synaptic silencing and the strengthening of GABA/glycinergic connections that normally occur with maturation. Functional mapping studies further revealed that these deficits markedly degraded the precision of tonotopy in this inhibitory auditory pathway. These results indicate that glutamate co-transmission is crucial for the synaptic reorganization and topographic specification of a developing inhibitory circuit.
Figures
Similar articles
-
Inhibitory synapses in the developing auditory system are glutamatergic.Nat Neurosci. 2005 Mar;8(3):332-8. doi: 10.1038/nn1397. Epub 2005 Jan 30. Nat Neurosci. 2005. PMID: 15746915
-
VGLUT3 does not synergize GABA/glycine release during functional refinement of an inhibitory auditory circuit.Front Neural Circuits. 2014 Nov 26;8:140. doi: 10.3389/fncir.2014.00140. eCollection 2014. Front Neural Circuits. 2014. PMID: 25505386 Free PMC article.
-
Maturation of calcium-dependent GABA, glycine, and glutamate release in the glycinergic MNTB-LSO pathway.PLoS One. 2013 Sep 19;8(9):e75688. doi: 10.1371/journal.pone.0075688. eCollection 2013. PLoS One. 2013. PMID: 24069436 Free PMC article.
-
Developmental Formation of the GABAergic and Glycinergic Networks in the Mouse Spinal Cord.Int J Mol Sci. 2022 Jan 13;23(2):834. doi: 10.3390/ijms23020834. Int J Mol Sci. 2022. PMID: 35055019 Free PMC article. Review.
-
Activity-dependent development of inhibitory synapses and innervation pattern: role of GABA signalling and beyond.J Physiol. 2009 May 1;587(Pt 9):1881-8. doi: 10.1113/jphysiol.2008.168211. Epub 2009 Feb 2. J Physiol. 2009. PMID: 19188247 Free PMC article. Review.
Cited by
-
Acupuncture effect and central autonomic regulation.Evid Based Complement Alternat Med. 2013;2013:267959. doi: 10.1155/2013/267959. Epub 2013 May 26. Evid Based Complement Alternat Med. 2013. PMID: 23762116 Free PMC article.
-
Glycinergic synaptic transmission in the cochlear nucleus of mice with normal hearing and age-related hearing loss.J Neurophysiol. 2013 Oct;110(8):1848-59. doi: 10.1152/jn.00151.2013. Epub 2013 Jul 31. J Neurophysiol. 2013. PMID: 23904491 Free PMC article.
-
Vesicular and plasma membrane transporters for neurotransmitters.Cold Spring Harb Perspect Biol. 2012 Feb 1;4(2):a005595. doi: 10.1101/cshperspect.a005595. Cold Spring Harb Perspect Biol. 2012. PMID: 22199021 Free PMC article. Review.
-
Auditory brainstem development and plasticity.Curr Opin Physiol. 2020 Dec;18:7-10. doi: 10.1016/j.cophys.2020.07.002. Epub 2020 Jul 7. Curr Opin Physiol. 2020. PMID: 33103017 Free PMC article.
-
Single-Cell Transcriptomic Reveals Dual and Multi-Transmitter Use in Neurons Across Metazoans.Front Mol Neurosci. 2021 Feb 1;14:623148. doi: 10.3389/fnmol.2021.623148. eCollection 2021. Front Mol Neurosci. 2021. PMID: 33597849 Free PMC article.
References
-
- Tollin DJ. The lateral superior olive: a functional role in sound source localization. Neuroscientist. 2003;9:127–143. - PubMed
-
- Kim G, Kandler K. Elimination and strengthening of glycinergic/GABAergic connections during tonotopic map formation. Nat Neurosci. 2003;6:282–290. - PubMed
-
- Nabekura J, et al. Developmental switch from GABA to glycine release in single central synaptic terminals. Nat Neurosci. 2004;7:17–23. - PubMed
-
- Gillespie DC, Kim G, Kandler K. Inhibitory synapses in the developing auditory system are glutamatergic. Nat Neurosci. 2005;8:332–338. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

