Serotonin 3A receptor subtype as an early and protracted marker of cortical interneuron subpopulations
- PMID: 20083553
- PMCID: PMC2936799
- DOI: 10.1093/cercor/bhp310
Serotonin 3A receptor subtype as an early and protracted marker of cortical interneuron subpopulations
Abstract
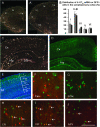
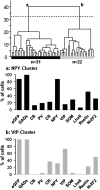
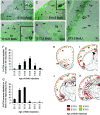
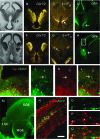
To identify neocortical neurons expressing the type 3 serotonergic receptor, here we used transgenic mice expressing the enhanced green fluorescent protein (GFP) under the control of the 5-HT(3A) promoter (5-HT(3A):GFP mice). By means of whole-cell patch-clamp recordings, biocytin labeling, and single-cell reversed-transcriptase polymerase chain reaction on acute brain slices of 5-HT(3A):GFP mice, we identified 2 populations of 5-HT(3A)-expressing interneurons within the somatosensory cortex. The first population was characterized by the frequent expression of the vasoactive intestinal peptide and a typical bipolar/bitufted morphology, whereas the second population expressed predominantly the neuropeptide Y and exhibited more complex dendritic arborizations. Most interneurons of this second group appeared very similar to neurogliaform cells according to their electrophysiological, molecular, and morphological properties. The combination of 5-bromo-2-deoxyuridine injections with 5-HT(3A) mRNA detection showed that cortical 5-HT(3A) interneurons are generated around embryonic day 14.5. Although at this stage the 5-HT(3A) receptor subunit is expressed in both the caudal ganglionic eminence and the entopeduncular area, homochronic in utero grafts experiments revealed that cortical 5-HT(3A) interneurons are mainly generated in the caudal ganglionic eminence. This protracted expression of the 5-HT(3A) subunit allowed us to study specific cortical interneuron populations from their birth to their final functional phenotype.
Figures

Similar articles
-
Dual origins of functionally distinct O-LM interneurons revealed by differential 5-HT(3A)R expression.Nat Neurosci. 2013 Nov;16(11):1598-607. doi: 10.1038/nn.3538. Epub 2013 Oct 6. Nat Neurosci. 2013. PMID: 24097043 Free PMC article.
-
The largest group of superficial neocortical GABAergic interneurons expresses ionotropic serotonin receptors.J Neurosci. 2010 Dec 15;30(50):16796-808. doi: 10.1523/JNEUROSCI.1869-10.2010. J Neurosci. 2010. PMID: 21159951 Free PMC article.
-
5-HT(3A) receptor-bearing white matter interstitial GABAergic interneurons are functionally integrated into cortical and subcortical networks.J Neurosci. 2011 Nov 16;31(46):16844-54. doi: 10.1523/JNEUROSCI.0310-11.2011. J Neurosci. 2011. PMID: 22090510 Free PMC article.
-
[Cortical 5-hydroxytryptamine receptor 3A (Htr3a) positive inhibitory neurons: diversity in type and function].Sheng Li Xue Bao. 2021 Apr 25;73(2):295-305. Sheng Li Xue Bao. 2021. PMID: 33903891 Review. Chinese.
-
The development of MGE-derived cortical interneurons: An Lhx6 tale.Int J Dev Biol. 2022;66(1-2-3):43-49. doi: 10.1387/ijdb.210185md. Int J Dev Biol. 2022. PMID: 34881792 Review.
Cited by
-
The serotonin 5-HT3 receptor: a novel neurodevelopmental target.Front Cell Neurosci. 2013 May 27;7:76. doi: 10.3389/fncel.2013.00076. eCollection 2013. Front Cell Neurosci. 2013. PMID: 23761731 Free PMC article.
-
Elevated serotonergic signaling amplifies synaptic noise and facilitates the emergence of epileptiform network oscillations.J Neurophysiol. 2014 Nov 15;112(10):2357-73. doi: 10.1152/jn.00031.2014. Epub 2014 Aug 13. J Neurophysiol. 2014. PMID: 25122717 Free PMC article.
-
Therapeutics of 5-HT3 receptor antagonists: current uses and future directions.Pharmacol Ther. 2011 Jun;130(3):338-47. doi: 10.1016/j.pharmthera.2011.02.003. Epub 2011 Feb 26. Pharmacol Ther. 2011. PMID: 21356241 Free PMC article. Review.
-
Neuronal nitric oxide synthase expressing neurons: a journey from birth to neuronal circuits.Front Neural Circuits. 2012 Dec 5;6:82. doi: 10.3389/fncir.2012.00082. eCollection 2012. Front Neural Circuits. 2012. PMID: 23227003 Free PMC article.
-
Rostro-Caudal and Caudo-Rostral Migrations in the Telencephalon: Going Forward or Backward?Front Neurosci. 2017 Dec 21;11:692. doi: 10.3389/fnins.2017.00692. eCollection 2017. Front Neurosci. 2017. PMID: 29311773 Free PMC article. Review.
References
-
- Anderson S, Mione M, Yun K, Rubenstein JL. Differential origins of neocortical projection and local circuit neurons: roles of Dlx genes in neocortical interneurogenesis. Cereb Cortex. 1999;9:646–654. - PubMed
-
- Anderson SA, Marín O, Horn C, Jennings K, Rubenstein JL. Distinct cortical migrations from the medial and lateral ganglionic eminences. Development. 2001;128:353–363. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

