GRASP55 and GRASP65 play complementary and essential roles in Golgi cisternal stacking
- PMID: 20083603
- PMCID: PMC2812519
- DOI: 10.1083/jcb.200907132
GRASP55 and GRASP65 play complementary and essential roles in Golgi cisternal stacking
Abstract
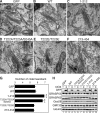
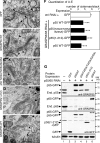
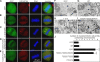
In vitro studies have suggested that Golgi stack formation involves two homologous peripheral Golgi proteins, GRASP65 and GRASP55, which localize to the cis and medial-trans cisternae, respectively. However, no mechanism has been provided on how these two GRASP proteins work together to stack Golgi cisternae. Here, we show that depletion of either GRASP55 or GRASP65 by siRNA reduces the number of cisternae per Golgi stack, whereas simultaneous knockdown of both GRASP proteins leads to disassembly of the entire stack. GRASP55 stacks Golgi membranes by forming oligomers through its N-terminal GRASP domain. This process is regulated by phosphorylation within the C-terminal serine/proline-rich domain. Expression of nonphosphorylatable GRASP55 mutants enhances Golgi stacking in interphase cells and inhibits Golgi disassembly during mitosis. These results demonstrate that GRASP55 and GRASP65 stack mammalian Golgi cisternae via a common mechanism.
Figures


Similar articles
-
The role of GRASP65 in Golgi cisternal stacking and cell cycle progression.Traffic. 2010 Jun;11(6):827-42. doi: 10.1111/j.1600-0854.2010.01055.x. Epub 2010 Feb 27. Traffic. 2010. PMID: 20214750 Free PMC article.
-
GRASP55, a second mammalian GRASP protein involved in the stacking of Golgi cisternae in a cell-free system.EMBO J. 1999 Sep 15;18(18):4949-60. doi: 10.1093/emboj/18.18.4949. EMBO J. 1999. PMID: 10487747 Free PMC article.
-
Knockout of the Golgi stacking proteins GRASP55 and GRASP65 impairs Golgi structure and function.Mol Biol Cell. 2017 Oct 15;28(21):2833-2842. doi: 10.1091/mbc.E17-02-0112. Epub 2017 Aug 16. Mol Biol Cell. 2017. PMID: 28814501 Free PMC article.
-
Nonredundant Roles of GRASP55 and GRASP65 in the Golgi Apparatus and Beyond.Trends Biochem Sci. 2020 Dec;45(12):1065-1079. doi: 10.1016/j.tibs.2020.08.001. Epub 2020 Sep 4. Trends Biochem Sci. 2020. PMID: 32893104 Free PMC article. Review.
-
GRASPs in Golgi Structure and Function.Front Cell Dev Biol. 2016 Jan 6;3:84. doi: 10.3389/fcell.2015.00084. eCollection 2015. Front Cell Dev Biol. 2016. PMID: 26779480 Free PMC article. Review.
Cited by
-
Mitotic inhibition of GRASP65 organelle tethering involves Polo-like kinase 1 (PLK1) phosphorylation proximate to an internal PDZ ligand.J Biol Chem. 2010 Dec 17;285(51):39994-40003. doi: 10.1074/jbc.M110.189449. Epub 2010 Oct 11. J Biol Chem. 2010. PMID: 20937827 Free PMC article.
-
Bridging the gap: membrane contact sites in signaling, metabolism, and organelle dynamics.J Cell Biol. 2014 Jun 23;205(6):759-69. doi: 10.1083/jcb.201401126. J Cell Biol. 2014. PMID: 24958771 Free PMC article.
-
Genetic, structural, and chemical insights into the dual function of GRASP55 in germ cell Golgi remodeling and JAM-C polarized localization during spermatogenesis.PLoS Genet. 2017 Jun 15;13(6):e1006803. doi: 10.1371/journal.pgen.1006803. eCollection 2017 Jun. PLoS Genet. 2017. PMID: 28617811 Free PMC article.
-
Retro-2 alters Golgi structure.Sci Rep. 2022 Sep 2;12(1):14975. doi: 10.1038/s41598-022-19415-x. Sci Rep. 2022. PMID: 36056100 Free PMC article.
-
Grasp55-/- mice display impaired fat absorption and resistance to high-fat diet-induced obesity.Nat Commun. 2020 Mar 17;11(1):1418. doi: 10.1038/s41467-020-14912-x. Nat Commun. 2020. PMID: 32184397 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

