Chronic network stimulation enhances evoked action potentials
- PMID: 20083862
- PMCID: PMC3775841
- DOI: 10.1088/1741-2560/7/1/016008
Chronic network stimulation enhances evoked action potentials
Abstract
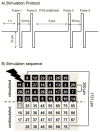
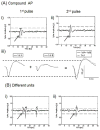
Neurons cultured on multielectrode arrays almost always lack external stimulation except during the acute experimental phase. We have investigated the effects of chronic stimulation during the course of development in cultured hippocampal neural networks by applying paired pulses at half of the electrodes for 0, 1 or 3 r/day for 8 days. Spike latencies increased from 4 to 16 ms as the distance from the stimulus increased from 200 to 1700 microm, suggesting an average of four synapses over this distance. Compared to no chronic stimulation, our results indicate that chronic stimulation increased evoked spike counts per stimulus by 50% at recording sites near the stimulating electrode and increased the instantaneous firing rate. On trials where both pulses elicited responses, spike count was 40-80% higher than when only one of the pulses elicited a response. In attempts to identify spike amplitude plasticity, we found mainly amplitude variation with different latencies suggesting recordings from neurons with different identities. These data suggest plastic network changes induced by chronic stimulation that enhance the reliability of information transmission and the efficiency of multisynaptic network communication.
Figures
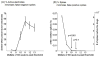








Similar articles
-
Chronic electrical stimulation of cultured hippocampal networks increases spontaneous spike rates.J Neurosci Methods. 2009 Oct 30;184(1):104-9. doi: 10.1016/j.jneumeth.2009.07.031. Epub 2009 Aug 8. J Neurosci Methods. 2009. PMID: 19666055 Free PMC article.
-
Properties of paired-pulse firing thresholds and the relationship with paired-pulse plasticity in hippocampal CA3-CA1 synapses.Eur J Neurosci. 2007 Jun;25(11):3253-63. doi: 10.1111/j.1460-9568.2007.05539.x. Eur J Neurosci. 2007. PMID: 17552994
-
Limbic gamma rhythms. II. Synaptic and intrinsic mechanisms underlying spike doublets in oscillating subicular neurons.J Neurophysiol. 1998 Jul;80(1):162-71. doi: 10.1152/jn.1998.80.1.162. J Neurophysiol. 1998. PMID: 9658038
-
Spontaneous activity and recurrent inhibition in cultured hippocampal networks.Synapse. 1993 Jul;14(3):206-13. doi: 10.1002/syn.890140304. Synapse. 1993. PMID: 8211707
-
Inhibitory synaptic plasticity regulates pyramidal neuron spiking in the rodent hippocampus.Neuroscience. 2008 Jul 31;155(1):64-75. doi: 10.1016/j.neuroscience.2008.05.009. Epub 2008 May 21. Neuroscience. 2008. PMID: 18562122
Cited by
-
Interaction of electrically evoked activity with intrinsic dynamics of cultured cortical networks with and without functional fast GABAergic synaptic transmission.Front Cell Neurosci. 2015 Jul 17;9:272. doi: 10.3389/fncel.2015.00272. eCollection 2015. Front Cell Neurosci. 2015. PMID: 26236196 Free PMC article.
-
Optogenetic stimulation effectively enhances intrinsically generated network synchrony.Front Neural Circuits. 2013 Oct 22;7:167. doi: 10.3389/fncir.2013.00167. eCollection 2013. Front Neural Circuits. 2013. PMID: 24155695 Free PMC article.
-
Electrical Responses and Spontaneous Activity of Human iPS-Derived Neuronal Networks Characterized for 3-month Culture with 4096-Electrode Arrays.Front Neurosci. 2016 Mar 30;10:121. doi: 10.3389/fnins.2016.00121. eCollection 2016. Front Neurosci. 2016. PMID: 27065786 Free PMC article.
-
Chronic stimulation of cultured neuronal networks boosts low-frequency oscillatory activity at theta and gamma with spikes phase-locked to gamma frequencies.J Neural Eng. 2012 Apr;9(2):026015. doi: 10.1088/1741-2560/9/2/026015. Epub 2012 Feb 23. J Neural Eng. 2012. PMID: 22361724 Free PMC article.
-
The Profile of Network Spontaneous Activity and Functional Organization Interplay in Hierarchically Connected Modular Neural Networks In Vitro.Micromachines (Basel). 2024 May 31;15(6):732. doi: 10.3390/mi15060732. Micromachines (Basel). 2024. PMID: 38930702 Free PMC article.
References
-
- Berdondini L, Massobrio P, Chiappalone M, Tedesco M, Imfeld K, Maccione A, Gandolfo M, Koudelka-Hep M, Martinoia S. Extracellular recordings from locally dense microelectrode arrays coupled to dissociated cortical cultures. J Neurosci Methods 2008 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
