Tri-partite complex for axonal transport drug delivery achieves pharmacological effect
- PMID: 20085661
- PMCID: PMC2837052
- DOI: 10.1186/1471-2202-11-8
Tri-partite complex for axonal transport drug delivery achieves pharmacological effect
Abstract
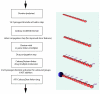
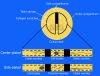
Background: Targeted delivery of pharmaceutical agents into selected populations of CNS (Central Nervous System) neurons is an extremely compelling goal. Currently, systemic methods are generally used for delivery of pain medications, anti-virals for treatment of dermatomal infections, anti-spasmodics, and neuroprotectants. Systemic side effects or undesirable effects on parts of the CNS that are not involved in the pathology limit efficacy and limit clinical utility for many classes of pharmaceuticals. Axonal transport from the periphery offers a possible selective route, but there has been little progress towards design of agents that can accomplish targeted delivery via this intraneural route. To achieve this goal, we developed a tripartite molecular construction concept involving an axonal transport facilitator molecule, a polymer linker, and a large number of drug molecules conjugated to the linker, then sought to evaluate its neurobiology and pharmacological behavior.
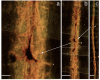
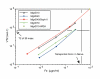
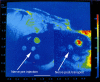
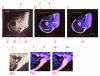
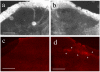
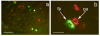
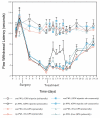
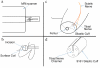
Results: We developed chemical synthesis methodologies for assembling these tripartite complexes using a variety of axonal transport facilitators including nerve growth factor, wheat germ agglutinin, and synthetic facilitators derived from phage display work. Loading of up to 100 drug molecules per complex was achieved. Conjugation methods were used that allowed the drugs to be released in active form inside the cell body after transport. Intramuscular and intradermal injection proved effective for introducing pharmacologically effective doses into selected populations of CNS neurons. Pharmacological efficacy with gabapentin in a paw withdrawal latency model revealed a ten fold increase in half life and a 300 fold decrease in necessary dose relative to systemic administration for gabapentin when the drug was delivered by axonal transport using the tripartite vehicle.
Conclusion: Specific targeting of selected subpopulations of CNS neurons for drug delivery by axonal transport holds great promise. The data shown here provide a basic framework for the intraneural pharmacology of this tripartite complex. The pharmacologically efficacious drug delivery demonstrated here verify the fundamental feasibility of using axonal transport for targeted drug delivery.
Figures









Similar articles
-
Gabapentin in acute postoperative pain management.Biomed Res Int. 2014;2014:631756. doi: 10.1155/2014/631756. Epub 2014 Apr 14. Biomed Res Int. 2014. PMID: 24829909 Free PMC article. Review.
-
Pharmacokinetic/Pharmacodynamic Relationship of Gabapentin in a CFA-induced Inflammatory Hyperalgesia Rat Model.Pharm Res. 2016 May;33(5):1133-43. doi: 10.1007/s11095-016-1859-7. Epub 2016 Jan 15. Pharm Res. 2016. PMID: 26780452
-
Nanosponge-based pediatric-controlled release dry suspension of Gabapentin for reconstitution.Drug Dev Ind Pharm. 2015;41(12):2029-36. doi: 10.3109/03639045.2015.1044903. Epub 2015 May 26. Drug Dev Ind Pharm. 2015. PMID: 26006328
-
Pharmacokinetics of oral gabapentin in greyhound dogs.Vet J. 2011 Jan;187(1):133-5. doi: 10.1016/j.tvjl.2009.09.022. Epub 2009 Oct 23. Vet J. 2011. PMID: 19854080 Free PMC article.
-
Clinical development of a once-daily gastroretentive formulation of gabapentin for treatment of postherpetic neuralgia: an overview.Expert Opin Drug Deliv. 2012 Sep;9(9):1147-60. doi: 10.1517/17425247.2012.709231. Epub 2012 Jul 18. Expert Opin Drug Deliv. 2012. PMID: 22809245 Review.
Cited by
-
Nanoparticle-mediated signaling endosome localization regulates growth cone motility and neurite growth.Proc Natl Acad Sci U S A. 2011 Nov 22;108(47):19042-7. doi: 10.1073/pnas.1019624108. Epub 2011 Nov 7. Proc Natl Acad Sci U S A. 2011. PMID: 22065745 Free PMC article.
-
Diffusion tensor imaging in hemorrhagic stroke.Exp Neurol. 2015 Oct;272:88-96. doi: 10.1016/j.expneurol.2015.05.011. Epub 2015 May 23. Exp Neurol. 2015. PMID: 26015333 Free PMC article. Review.
-
MR Imaging of Peripheral Nerves Using Targeted Application of Contrast Agents: An Experimental Proof-of-Concept Study.Front Med (Lausanne). 2020 Dec 11;7:613138. doi: 10.3389/fmed.2020.613138. eCollection 2020. Front Med (Lausanne). 2020. PMID: 33363189 Free PMC article.
-
Nanotransducers for Wireless Neuromodulation.Matter. 2021 May 5;4(5):1484-1510. doi: 10.1016/j.matt.2021.02.012. Matter. 2021. PMID: 33997768 Free PMC article.
-
Magnetic resonance neurography and diffusion tensor imaging: origins, history, and clinical impact of the first 50,000 cases with an assessment of efficacy and utility in a prospective 5000-patient study group.Neurosurgery. 2009 Oct;65(4 Suppl):A29-43. doi: 10.1227/01.NEU.0000351279.78110.00. Neurosurgery. 2009. PMID: 19927075 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

