Working with OpusXpress: methods for high volume oocyte experiments
- PMID: 20085813
- PMCID: PMC2886986
- DOI: 10.1016/j.ymeth.2010.01.012
Working with OpusXpress: methods for high volume oocyte experiments
Abstract
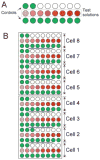
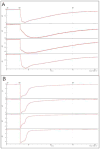
OpusXpress is a semi-automated system for high throughput voltage clamp recording from Xenopus oocytes. We participated in the development process for this system and were the only laboratory to field test a prototype. Subsequently, we obtained an early production model that we have used on a regular basis for the last seven years, conducting many thousands of experiments, publishing extensively, and carrying out collaborative research in drug discovery. In this article, we relate our experience with the OpusXpress recording system and large volume oocyte handling. We provide our standard operating procedures and outline the organization of our successful team. Some of our advice is specific to researchers fortunate enough to have access to an OpusXpress system, but most of it is applicable to any group using Xenopus oocytes for the heterologous expression of ion channels.
Figures



Similar articles
-
High throughput electrophysiology with Xenopus oocytes.Comb Chem High Throughput Screen. 2009 Jan;12(1):38-50. doi: 10.2174/138620709787047975. Comb Chem High Throughput Screen. 2009. PMID: 19149490 Free PMC article. Review.
-
Xenopus oocytes as a heterologous expression system for studying ion channels with the patch-clamp technique.Methods Mol Biol. 2008;491:127-39. doi: 10.1007/978-1-59745-526-8_10. Methods Mol Biol. 2008. PMID: 18998089
-
Cave Canalem: how endogenous ion channels may interfere with heterologous expression in Xenopus oocytes.Methods. 2010 May;51(1):66-74. doi: 10.1016/j.ymeth.2010.01.034. Epub 2010 Feb 1. Methods. 2010. PMID: 20123125 Review.
-
Voltage clamp recordings from Xenopus oocytes.Curr Protoc Neurosci. 2001 May;Chapter 6:Unit 6.12. doi: 10.1002/0471142301.ns0612s10. Curr Protoc Neurosci. 2001. PMID: 18428511
-
P2X Electrophysiology and Surface Trafficking in Xenopus Oocytes.Methods Mol Biol. 2020;2041:243-259. doi: 10.1007/978-1-4939-9717-6_18. Methods Mol Biol. 2020. PMID: 31646494
Cited by
-
Evidence for Two Modes of Binding of the Negative Allosteric Modulator SB269,652 to the Dopamine D2 Receptor.Biomedicines. 2021 Dec 23;10(1):22. doi: 10.3390/biomedicines10010022. Biomedicines. 2021. PMID: 35052702 Free PMC article.
-
Allosteric Agonism of α7 Nicotinic Acetylcholine Receptors: Receptor Modulation Outside the Orthosteric Site.Mol Pharmacol. 2019 Jun;95(6):606-614. doi: 10.1124/mol.119.115758. Epub 2019 Apr 3. Mol Pharmacol. 2019. PMID: 30944209 Free PMC article.
-
Insights Into the Differential Desensitization of α4β2 Nicotinic Acetylcholine Receptor Isoforms Obtained With Positive Allosteric Modulation of Mutant Receptors.Mol Pharmacol. 2023 Feb;103(2):63-76. doi: 10.1124/molpharm.122.000591. Epub 2022 Nov 22. Mol Pharmacol. 2023. PMID: 36414373 Free PMC article.
-
Estimation of Dopamine D1 Receptor Agonist Binding Kinetics Using Time-Resolved Functional Assays: Relation to Agonist-Induced Receptor Internalization by Investigational Antiparkinsonian Therapeutics.ACS Chem Neurosci. 2025 Jul 2;16(13):2502-2512. doi: 10.1021/acschemneuro.5c00270. Epub 2025 Jun 19. ACS Chem Neurosci. 2025. PMID: 40537032 Free PMC article.
-
Sulfonium Ligands of the α7 nAChR.Molecules. 2021 Sep 17;26(18):5643. doi: 10.3390/molecules26185643. Molecules. 2021. PMID: 34577114 Free PMC article.
References
-
- Bossi E, Fabbrini MS, Ceriotti A. Methods Mol Biol. 2007;375:107–31. - PubMed
-
- Morera FJ, Vargas G, Gonzalez C, et al. Methods Mol Biol. 2007;400:571–85. - PubMed
-
- Sigel E, Minier F. Mol Nutr Food Res. 2005;49:228–34. - PubMed
-
- Methfessel C, Witzemann V, Takahashi T, et al. Pflüegers Arch Eur J Physiol. 1986;407:577–588. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

