E-cadherin differentially regulates the assembly of Connexin43 and Connexin32 into gap junctions in human squamous carcinoma cells
- PMID: 20086013
- PMCID: PMC2856283
- DOI: 10.1074/jbc.M109.053348
E-cadherin differentially regulates the assembly of Connexin43 and Connexin32 into gap junctions in human squamous carcinoma cells
Abstract
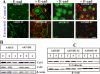
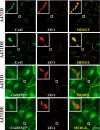
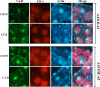
It is as yet unknown how the assembly of connexins (Cx) into gap junctions (GJ) is initiated upon cell-cell contact. We investigated whether the trafficking and assembly of Cx43 and Cx32 into GJs were contingent upon cell-cell adhesion mediated by E-cadherin. We also examined the role of the carboxyl termini of these Cxs in initiating the formation of GJs. Using cadherin and Cx-null cells, and by introducing Cx43 and Cx32, either alone or in combination with E-cadherin, our studies demonstrated that E-cadherin-mediated cell-cell adhesion was neither essential nor sufficient to initiate GJ assembly de novo in A431D human squamous carcinoma cells. However, E-cadherin facilitated the growth and assembly of preformed GJs composed of Cx43, although the growth of cells on Transwell filters was required to initiate the assembly of Cx32. Our results also documented that the carboxyl termini of both Cxs were required in this cell type to initiate the formation of GJs de novo. Our findings also showed that GJ puncta composed of Cx43 co-localized extensively with ZO-1 and actin fibers at cell peripheries and that ZO-1 knockdown attenuated Cx43 assembly. These findings suggest that the assembly of Cx43 and Cx32 into GJs is differentially modulated by E-cadherin-mediated cell-cell adhesion and that direct or indirect cross-talk between carboxyl tails of Cxs and actin cytoskeleton via ZO-1 may regulate GJ assembly and growth.
Figures







Similar articles
-
Assembly of connexin43 into gap junctions is regulated differentially by E-cadherin and N-cadherin in rat liver epithelial cells.Mol Biol Cell. 2010 Dec;21(23):4089-107. doi: 10.1091/mbc.E10-05-0403. Epub 2010 Sep 29. Mol Biol Cell. 2010. PMID: 20881055 Free PMC article.
-
Alterations in Cx43 and OB-cadherin affect breast cancer cell metastatic potential.Clin Exp Metastasis. 2008;25(3):265-72. doi: 10.1007/s10585-007-9140-4. Epub 2008 Jan 10. Clin Exp Metastasis. 2008. PMID: 18193170
-
Connexin 43 connexon to gap junction transition is regulated by zonula occludens-1.Mol Biol Cell. 2011 May;22(9):1516-28. doi: 10.1091/mbc.E10-06-0548. Epub 2011 Mar 16. Mol Biol Cell. 2011. PMID: 21411628 Free PMC article.
-
Role of gap junctions in lung neoplasia.Exp Lung Res. 1998 Jul-Aug;24(4):523-39. doi: 10.3109/01902149809087384. Exp Lung Res. 1998. PMID: 9659581 Review.
-
The unstoppable connexin43 carboxyl-terminus: new roles in gap junction organization and wound healing.Ann N Y Acad Sci. 2006 Oct;1080:49-62. doi: 10.1196/annals.1380.005. Ann N Y Acad Sci. 2006. PMID: 17132774 Review.
Cited by
-
The Multifaceted Role of Connexins in Tumor Microenvironment Initiation and Maintenance.Biology (Basel). 2023 Jan 28;12(2):204. doi: 10.3390/biology12020204. Biology (Basel). 2023. PMID: 36829482 Free PMC article. Review.
-
Dileucine-like motifs in the C-terminal tail of connexin32 control its endocytosis and assembly into gap junctions.J Cell Sci. 2018 Mar 2;131(5):jcs207340. doi: 10.1242/jcs.207340. J Cell Sci. 2018. PMID: 29361528 Free PMC article.
-
Assembly of connexin43 into gap junctions is regulated differentially by E-cadherin and N-cadherin in rat liver epithelial cells.Mol Biol Cell. 2010 Dec;21(23):4089-107. doi: 10.1091/mbc.E10-05-0403. Epub 2010 Sep 29. Mol Biol Cell. 2010. PMID: 20881055 Free PMC article.
-
Characterization of the structure and intermolecular interactions between the connexin 32 carboxyl-terminal domain and the protein partners synapse-associated protein 97 and calmodulin.J Biol Chem. 2012 Aug 10;287(33):27771-88. doi: 10.1074/jbc.M112.382572. Epub 2012 Jun 20. J Biol Chem. 2012. PMID: 22718765 Free PMC article.
-
The role of connexins in breast cancer: from misregulated cell communication to aberrant intracellular signaling.Tissue Barriers. 2022 Jan 2;10(1):1962698. doi: 10.1080/21688370.2021.1962698. Epub 2021 Aug 6. Tissue Barriers. 2022. PMID: 34355641 Free PMC article. Review.
References
-
- Beyer E. C., Berthoud V. M. (2009) in Connexins: A Guide (Harris A., Locke D. eds) pp. 3–26, Springer, Humana Press, NY
-
- Sosinsky G. E., Nicholson B. J. (2005) Biochim. Biophys. Acta 1711, 99–125 - PubMed
-
- Gaietta G., Deerinck T. J., Adams S. R., Bouwer J., Tour O., Laird D. W., Sosinsky G. E., Tsien R. Y., Ellisman M. H. (2002) Science 296, 503–507 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

