A novel geranylgeranyl transferase inhibitor in combination with lovastatin inhibits proliferation and induces autophagy in STS-26T MPNST cells
- PMID: 20086055
- PMCID: PMC2846025
- DOI: 10.1124/jpet.109.160192
A novel geranylgeranyl transferase inhibitor in combination with lovastatin inhibits proliferation and induces autophagy in STS-26T MPNST cells
Abstract
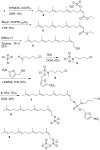
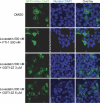
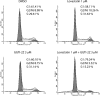
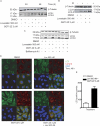
Prenylation inhibitors have gained increasing attention as potential therapeutics for cancer. Initial work focused on inhibitors of farnesylation, but more recently geranylgeranyl transferase inhibitors (GGTIs) have begun to be evaluated for their potential antitumor activity in vitro and in vivo. In this study, we have developed a nonpeptidomimetic GGTI, termed GGTI-2Z [(5-nitrofuran-2-yl)methyl-(2Z,6E,10E)-3,7,11,15-tetramethylhexadeca-2,6,10,14-tetraenyl 4-chlorobutyl(methyl)phosphoramidate], which in combination with lovastatin inhibits geranylgeranyl transferase I (GGTase I) and GGTase II/RabGGTase, without affecting farnesylation. The combination treatment results in a G(0)/G(1) arrest and synergistic inhibition of proliferation of cultured STS-26T malignant peripheral nerve sheath tumor cells. We also show that the antiproliferative activity of drugs in combination occurs in the context of autophagy. The combination treatment also induces autophagy in the MCF10.DCIS model of human breast ductal carcinoma in situ and in 1c1c7 murine hepatoma cells, where it also reduces proliferation. At the same time, there is no detectable toxicity in normal immortalized Schwann cells. These studies establish GGTI-2Z as a novel geranylgeranyl pyrophosphate derivative that may work through a new mechanism involving the induction of autophagy and, in combination with lovastatin, may serve as a valuable paradigm for developing more effective strategies in this class of antitumor therapeutics.
Figures



Similar articles
-
Evaluation of farnesyl:protein transferase and geranylgeranyl:protein transferase inhibitor combinations in preclinical models.Cancer Res. 2001 Dec 15;61(24):8758-68. Cancer Res. 2001. PMID: 11751396
-
The geranylgeranyltransferase-I inhibitor GGTI-298 arrests human tumor cells in G0/G1 and induces p21(WAF1/CIP1/SDI1) in a p53-independent manner.J Biol Chem. 1997 Oct 24;272(43):27224-9. doi: 10.1074/jbc.272.43.27224. J Biol Chem. 1997. PMID: 9341167
-
Protein geranylgeranylation, not farnesylation, is required for the G1 to S phase transition in mouse fibroblasts.Oncogene. 1996 Nov 7;13(9):1991-9. Oncogene. 1996. PMID: 8934546
-
Farnesyltransferase and geranylgeranyltransferase I inhibitors and cancer therapy: lessons from mechanism and bench-to-bedside translational studies.Oncogene. 2000 Dec 27;19(56):6584-93. doi: 10.1038/sj.onc.1204146. Oncogene. 2000. PMID: 11426643 Review.
-
The mevalonate synthesis pathway as a therapeutic target in cancer.Clin Orthop Relat Res. 2003 Oct;(415 Suppl):S59-66. doi: 10.1097/01.blo.0000093846.72468.66. Clin Orthop Relat Res. 2003. PMID: 14600593 Review.
Cited by
-
Impaired Pten expression in human malignant peripheral nerve sheath tumours.PLoS One. 2012;7(11):e47595. doi: 10.1371/journal.pone.0047595. Epub 2012 Nov 6. PLoS One. 2012. PMID: 23139750 Free PMC article.
-
CD55 upregulation in astrocytes by statins as potential therapy for AQP4-IgG seropositive neuromyelitis optica.J Neuroinflammation. 2019 Mar 9;16(1):57. doi: 10.1186/s12974-019-1448-x. J Neuroinflammation. 2019. PMID: 30851734 Free PMC article.
-
Activated Ras as a Therapeutic Target: Constraints on Directly Targeting Ras Isoforms and Wild-Type versus Mutated Proteins.ISRN Oncol. 2013 Oct 31;2013:536529. doi: 10.1155/2013/536529. eCollection 2013. ISRN Oncol. 2013. PMID: 24294527 Free PMC article. Review.
-
Intratumoral injection of schwannoma with attenuated Salmonella typhimurium induces antitumor immunity and controls tumor growth.Proc Natl Acad Sci U S A. 2022 Jun 14;119(24):e2202719119. doi: 10.1073/pnas.2202719119. Epub 2022 Jun 8. Proc Natl Acad Sci U S A. 2022. PMID: 35675425 Free PMC article.
-
Prodrugs of phosphonates and phosphates: crossing the membrane barrier.Top Curr Chem. 2015;360:115-60. doi: 10.1007/128_2014_561. Top Curr Chem. 2015. PMID: 25391982 Free PMC article. Review.
References
-
- Apel A, Zentgraf H, Buchler MW, Herr I. (2009) Autophagy-a double-edged sword in oncology. Int J Cancer 125:991–995 - PubMed
-
- Araki M, Motojima K. (2008) Hydrophobic statins induce autophagy in cultured human rhabdomyosarcoma cells. Biochem Biophys Res Commun 367:462–467 - PubMed
-
- Bucci C, Parton RG, Mather IH, Stunnenberg H, Simons K, Hoflack B, Zerial M. (1992) The small GTPase rab5 functions as a regulatory factor in the early endocytic pathway. Cell 70:715–728 - PubMed
-
- Cheng KW, Lahad JP, Kuo WL, Lapuk A, Yamada K, Auersperg N, Liu J, Smith-McCune K, Lu KH, Fishman D, et al. (2004) The RAB25 small GTPase determines aggressiveness of ovarian and breast cancers. Nat Med 10:1251–1256 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

