New susceptibility breakpoints for first-line antituberculosis drugs based on antimicrobial pharmacokinetic/pharmacodynamic science and population pharmacokinetic variability
- PMID: 20086150
- PMCID: PMC2849358
- DOI: 10.1128/AAC.01474-09
New susceptibility breakpoints for first-line antituberculosis drugs based on antimicrobial pharmacokinetic/pharmacodynamic science and population pharmacokinetic variability
Abstract
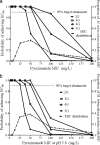
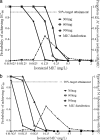
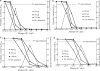
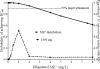
Arguably, one of the most common and consequential laboratory tests performed in the world is Mycobacterium tuberculosis susceptibility testing. M. tuberculosis resistance is defined by growth of > or =1% of a bacillary inoculum on the critical concentration of an antibiotic. The critical concentration was chosen based on inhibition of > or =95% of wild-type isolates. The critical concentration of isoniazid is either 0.2 or 1.0 mg/liter, that of rifampin is 1.0 mg/liter, that of pyrazinamide is 100 mg/liter, that of ethambutol is 5.0 mg/liter, and that of fluoroquinolones is 1.0 mg/liter. However, the relevance of these concentrations to microbiologic and clinical outcomes is unclear. Critical concentrations were identified using the ability to achieve the antibiotic area under the concentration-time curve/MIC ratio associated with > or =90% of maximal kill (EC(90)) of M. tuberculosis in > or =90% of patients. Population pharmacokinetic parameters and their variability encountered in tuberculosis patients were utilized in Monte Carlo simulations to determine the probability that particular daily doses of the drugs would achieve or exceed the EC(90) in the epithelial lining fluid of 10,000 tuberculosis patients. Failure to achieve EC(90) in > or =90% of patients at a particular MIC was defined as drug resistance. The critical concentrations of moxifloxacin and ethambutol remained unchanged, but a critical concentration of 50 mg/liter was identified for pyrazinamide, 0.0312 mg/liter and 0.125 mg/liter were defined for low- and high-level isoniazid resistance, respectively, and 0.0625 mg/liter was defined for rifampin. Thus, current critical concentrations of first-line antituberculosis drugs are overoptimistic and should be set lower. With the proposed breakpoints, the rates of multidrug-resistant tuberculosis could become 4-fold higher than currently assumed.
Figures

References
-
- Allison, S. T.1956. Pyrazinamide-isoniazid in low dosage in treatment of pulmonary tuberculosis. Am. Rev. Tuberc. 74:400-409. - PubMed
-
- Ambrose, P. G., S. M. Bhavnani, C. M. Rubino, A. Louie, T. Gumbo, A. Forrest, and G. L. Drusano.2007. Pharmacokinetics-pharmacodynamics of antimicrobial therapy: it's not just for mice anymore. Clin. Infect. Dis. 44:79-86. - PubMed
-
- Ambrose, P. G., A. K. Meagher, J. A. Passarell, S. A. Van Wart, B. B. Cirincione, S. M. Bhavnani, and E. Ellis-Grosse.2009. Application of patient population-derived pharmacokinetic-pharmacodynamic relationships to tigecycline breakpoint determination for staphylococci and streptococci. Diagn. Microbiol. Infect. Dis. 63:155-159. - PubMed
-
- Anonymous.2009. Anti-tuberculosis drug resistance in the world. Fourth global report. WHO/IUATLD Global Project on Anti-Tuberculosis Drug Resistance Surveillance, 2000-2007. World Health Organization, Geneva, Switzerland.
-
- Bidstrup, C., P. H. Andersen, P. Skinhoj, and A. B. Andersen.2002. Tuberculous meningitis in a country with a low incidence of tuberculosis: still a serious disease and a diagnostic challenge. Scand. J. Infect. Dis. 34:811-814. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

