Neutrophil gelatinase-associated lipocalin expresses antimicrobial activity by interfering with L-norepinephrine-mediated bacterial iron acquisition
- PMID: 20086155
- PMCID: PMC2849382
- DOI: 10.1128/AAC.01158-09
Neutrophil gelatinase-associated lipocalin expresses antimicrobial activity by interfering with L-norepinephrine-mediated bacterial iron acquisition
Abstract
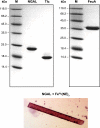
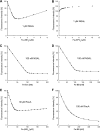
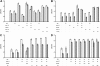
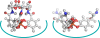
l-norepinephrine (NE) is a neuroendocrine catecholamine that supports bacterial growth by mobilizing iron from a primary source such as holotransferrin to increase its bioavailability for cellular uptake. Iron complexes of NE resemble those of bacterial siderophores that are scavenged by human neutrophil gelatinase-associated lipocalin (NGAL) as part of the innate immune defense. Here, we show that NGAL binds iron-complexed NE, indicating physiological relevance for both bacterial and human iron metabolism. The fluorescence titration of purified recombinant NGAL with the Fe(III).(NE)(3) iron complex revealed high affinity for this ligand, with a K(D) of 50.6 nM. In contrast, the binding protein FeuA of Bacillus subtilis, which is involved in the bacterial uptake of triscatecholate iron complexes, has a K(D) for Fe(III).(NE)(3) of 1.6 muM, indicating that NGAL is an efficient competitor. Furthermore, NGAL was shown to inhibit the NE-mediated growth of both E. coli and B. subtilis strains that either are capable or incapable of producing their native siderophores enterobactin and bacillibactin, respectively. These experiments suggest that iron-complexed NE directly serves as an iron source for bacterial uptake systems, and that NGAL can function as an antagonist of this iron acquisition process. Interestingly, a functional FeuABC uptake system was shown to be necessary for NE-mediated growth stimulation as well as its NGAL-dependent inhibition. This study demonstrates for the first time that human NGAL not only neutralizes pathogen-derived virulence factors but also can effectively scavenge an iron-chelate complex abundant in the host.
Figures
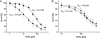
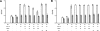
Similar articles
-
The neutrophil lipocalin NGAL is a bacteriostatic agent that interferes with siderophore-mediated iron acquisition.Mol Cell. 2002 Nov;10(5):1033-43. doi: 10.1016/s1097-2765(02)00708-6. Mol Cell. 2002. PMID: 12453412
-
The multifaceted roles of neutrophil gelatinase associated lipocalin (NGAL) in inflammation and cancer.Biochim Biophys Acta. 2012 Aug;1826(1):129-69. doi: 10.1016/j.bbcan.2012.03.008. Epub 2012 Mar 31. Biochim Biophys Acta. 2012. PMID: 22513004 Free PMC article. Review.
-
Neutrophil gelatinase-associated lipocalin-mediated iron traffic in kidney epithelia.Curr Opin Nephrol Hypertens. 2006 Jul;15(4):442-9. doi: 10.1097/01.mnh.0000232886.81142.58. Curr Opin Nephrol Hypertens. 2006. PMID: 16775460 Review.
-
Neutrophil gelatinase-associated lipocalin and innate immune responses to bacterial infections.Med Microbiol Immunol. 2015 Aug;204(4):471-9. doi: 10.1007/s00430-015-0394-1. Epub 2015 Feb 26. Med Microbiol Immunol. 2015. PMID: 25716557 Review.
-
Neutrophil gelatinase-associated lipocalin, a siderophore-binding eukaryotic protein.Biometals. 2006 Apr;19(2):211-5. doi: 10.1007/s10534-005-3251-7. Biometals. 2006. PMID: 16718606 Review.
Cited by
-
Bacteria in an intense competition for iron: Key component of the Campylobacter jejuni iron uptake system scavenges enterobactin hydrolysis product.Proc Natl Acad Sci U S A. 2016 May 24;113(21):5850-5. doi: 10.1073/pnas.1520829113. Epub 2016 May 9. Proc Natl Acad Sci U S A. 2016. PMID: 27162326 Free PMC article.
-
Astrocyte-Secreted Lcn2 Modulates Dendritic Spine Morphology.Cells. 2025 Jan 21;14(3):159. doi: 10.3390/cells14030159. Cells. 2025. PMID: 39936951 Free PMC article.
-
Parsing the functional specificity of Siderocalin/Lipocalin 2/NGAL for siderophores and related small-molecule ligands.J Struct Biol X. 2019 May 30;2:100008. doi: 10.1016/j.yjsbx.2019.100008. eCollection 2019 Apr-Jun. J Struct Biol X. 2019. PMID: 32647813 Free PMC article.
-
Klebsiella pneumoniae yersiniabactin promotes respiratory tract infection through evasion of lipocalin 2.Infect Immun. 2011 Aug;79(8):3309-16. doi: 10.1128/IAI.05114-11. Epub 2011 May 16. Infect Immun. 2011. PMID: 21576334 Free PMC article.
-
Interkingdom Cross-Talk in Times of Stress: Salmonella Typhimurium Grown in the Presence of Catecholamines Inhibits Porcine Immune Functionality in vitro.Front Immunol. 2020 Sep 30;11:572056. doi: 10.3389/fimmu.2020.572056. eCollection 2020. Front Immunol. 2020. PMID: 33101292 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

