Proapoptotic kinase MST2 coordinates signaling crosstalk between RASSF1A, Raf-1, and Akt
- PMID: 20086174
- PMCID: PMC2880716
- DOI: 10.1158/0008-5472.CAN-09-3147
Proapoptotic kinase MST2 coordinates signaling crosstalk between RASSF1A, Raf-1, and Akt
Abstract
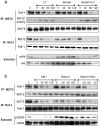
Mammalian MST kinases function in stress-induced apoptosis to limit tumor progression. However, there is limited understanding about MST2 control by key regulators of cell division and survival. Raf-1 binds and inhibits MST2 kinase, whereas dissociation from Raf-1 and binding to tumor suppressor protein RASSF1A activates MST2. Akt phosphorylates MST2 in response to mitogens, oncogenic Ras, or depletion of tumor suppressor phosphatase and tensin homologue deleted on chromosome 10. We identified T117 and T384 as Akt phosphorylation sites in MST2. Mutation of these sites inhibited MST2 binding to Raf-1 kinase but enhanced binding to tumor suppressor RASSF1A, accentuating downstream c-Jun NH(2)-terminal kinase and p38 mitogen-activated protein kinase signaling and promoting apoptosis. We determined that MST2 phosphorylation by Akt limits MST2 activity in two ways: first, by blocking its binding to RASSF1A and by promoting its association into the Raf-1 inhibitory complex, and second, by preventing homodimerization of MST2, which is needed for its activation. Dissociation of the Raf-1-MST2 complex promoted mitogenic signaling and coordinately licensed apoptotic risk. Using Ras effector domain mutants, we found that Akt is essential to prevent MST2 activation after mitogenic stimulation. Our findings elucidate how MST2 serves as a hub to integrate biological outputs of the Raf-1 and Akt pathways.
Figures




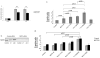
Similar articles
-
RASSF1A elicits apoptosis through an MST2 pathway directing proapoptotic transcription by the p73 tumor suppressor protein.Mol Cell. 2007 Sep 21;27(6):962-75. doi: 10.1016/j.molcel.2007.08.008. Mol Cell. 2007. PMID: 17889669 Free PMC article.
-
Taming the Hippo: Raf-1 controls apoptosis by suppressing MST2/Hippo.Cell Cycle. 2005 Mar;4(3):365-7. doi: 10.4161/cc.4.3.1531. Epub 2005 Mar 8. Cell Cycle. 2005. PMID: 15701972 Review.
-
Role of the kinase MST2 in suppression of apoptosis by the proto-oncogene product Raf-1.Science. 2004 Dec 24;306(5705):2267-70. doi: 10.1126/science.1103233. Science. 2004. PMID: 15618521
-
Mammalian Ste20-like kinase (Mst2) indirectly supports Raf-1/ERK pathway activity via maintenance of protein phosphatase-2A catalytic subunit levels and consequent suppression of inhibitory Raf-1 phosphorylation.J Biol Chem. 2010 May 14;285(20):15076-15087. doi: 10.1074/jbc.M109.078915. Epub 2010 Mar 8. J Biol Chem. 2010. PMID: 20212043 Free PMC article.
-
Mammalian sterile 20-like kinases in tumor suppression: an emerging pathway.Cancer Res. 2005 Jul 1;65(13):5485-7. doi: 10.1158/0008-5472.CAN-05-1453. Cancer Res. 2005. PMID: 15994916 Review.
Cited by
-
MST1/MST2 Protein Kinases: Regulation and Physiologic Roles.Biochemistry. 2016 Oct 4;55(39):5507-5519. doi: 10.1021/acs.biochem.6b00763. Epub 2016 Sep 26. Biochemistry. 2016. PMID: 27618557 Free PMC article. Review.
-
Pumping the brakes on RAS - negative regulators and death effectors of RAS.J Cell Sci. 2020 Feb 10;133(3):jcs238865. doi: 10.1242/jcs.238865. J Cell Sci. 2020. PMID: 32041893 Free PMC article. Review.
-
Protein kinases of the Hippo pathway: regulation and substrates.Semin Cell Dev Biol. 2012 Sep;23(7):770-84. doi: 10.1016/j.semcdb.2012.07.002. Epub 2012 Aug 9. Semin Cell Dev Biol. 2012. PMID: 22898666 Free PMC article. Review.
-
Competing to coordinate cell fate decisions: the MST2-Raf-1 signaling device.Cell Cycle. 2015;14(2):189-99. doi: 10.4161/15384101.2014.973743. Cell Cycle. 2015. PMID: 25607644 Free PMC article.
-
Noncanonical HIPPO/MST Signaling via BUB3 and FOXO Drives Pulmonary Vascular Cell Growth and Survival.Circ Res. 2022 Mar 4;130(5):760-778. doi: 10.1161/CIRCRESAHA.121.319100. Epub 2022 Feb 7. Circ Res. 2022. PMID: 35124974 Free PMC article.
References
-
- Ura S, Masuyama N, Graves JD, Gotoh Y. MST1-JNK promotes apoptosis via caspase-dependent and independent pathways. Genes Cells. 2001;6:519–30. - PubMed
-
- Lehtinen MK, Yuan Z, Boag PR, et al. A conserved MST-FOXO signaling pathway mediates oxidative-stress responses and extends life span. Cell. 2006;125:987–1001. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

