Anucleate platelets generate progeny
- PMID: 20086251
- PMCID: PMC2865870
- DOI: 10.1182/blood-2009-08-239558
Anucleate platelets generate progeny
Abstract
Platelets are classified as terminally differentiated cells that are incapable of cellular division. However, we observe that anucleate human platelets, either maintained in suspension culture or captured in microdrops, give rise to new cell bodies packed with respiring mitochondria and alpha-granules. Platelet progeny formation also occurs in whole blood cultures. Newly formed platelets are structurally indistinguishable from normal platelets, are able to adhere and spread on extracellular matrix, and display normal signal-dependent expression of surface P-selectin and annexin V. Platelet progeny formation is accompanied by increases in biomass, cellular protein levels, and protein synthesis in expanding populations. Platelet numbers also increase during ex vivo storage. These observations indicate that platelets have a previously unrecognized capacity for producing functional progeny, which involves a form of cell division that does not require a nucleus. Because this new function of platelets occurs outside of the bone marrow milieu, it raises the possibility that thrombopoiesis continues in the bloodstream.
Figures
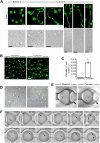
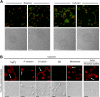
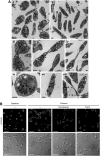
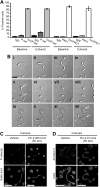
Comment in
-
Platelets in bloom.Blood. 2010 May 6;115(18):3650-1. doi: 10.1182/blood-2010-02-268714. Blood. 2010. PMID: 20448116 No abstract available.
Similar articles
-
Platelet alpha granule deficiency associated with decreased P-selectin and selective impairment of thrombin-induced activation in a new patient with gray platelet syndrome (alpha-storage pool deficiency).J Lab Clin Med. 1997 Mar;129(3):364-75. doi: 10.1016/s0022-2143(97)90185-2. J Lab Clin Med. 1997. PMID: 9042822
-
Tissue factor-enriched vesicles are taken up by platelets and induce platelet aggregation in the presence of factor VIIa.Thromb Haemost. 2007 Feb;97(2):202-11. Thromb Haemost. 2007. PMID: 17264948
-
Role of FcRgamma and factor XIIIA in coated platelet formation.Blood. 2005 Dec 15;106(13):4146-51. doi: 10.1182/blood-2005-03-1223. Epub 2005 Aug 16. Blood. 2005. PMID: 16105983 Free PMC article.
-
[Platelet structure and functions (a review of literature). Part 1].Klin Lab Diagn. 2010 Nov;(11):21-6. Klin Lab Diagn. 2010. PMID: 21313750 Review. Russian.
-
Apoptosis in the anucleate platelet.Blood Rev. 2012 Mar;26(2):51-63. doi: 10.1016/j.blre.2011.10.002. Epub 2011 Nov 4. Blood Rev. 2012. PMID: 22055392 Review.
Cited by
-
Autophagy and its consequences for platelet biology.Thromb Res. 2023 Nov;231:170-181. doi: 10.1016/j.thromres.2022.08.019. Epub 2022 Aug 27. Thromb Res. 2023. PMID: 36058760 Free PMC article. Review.
-
Role of microparticles in dengue virus infection and its impact on medical intervention strategies.Yale J Biol Med. 2012 Mar;85(1):3-18. Epub 2012 Mar 29. Yale J Biol Med. 2012. PMID: 22461739 Free PMC article. Review.
-
A New Role of NAP1L1 in Megakaryocytes and Human Platelets.Int J Mol Sci. 2022 Nov 24;23(23):14694. doi: 10.3390/ijms232314694. Int J Mol Sci. 2022. PMID: 36499021 Free PMC article.
-
Direct Cryo-ET observation of platelet deformation induced by SARS-CoV-2 spike protein.Nat Commun. 2023 Feb 4;14(1):620. doi: 10.1038/s41467-023-36279-5. Nat Commun. 2023. PMID: 36739444 Free PMC article.
-
RGDfK-functionalized gold nanorods bind only to activated platelets.J Biomed Mater Res A. 2017 Jan;105(1):209-217. doi: 10.1002/jbm.a.35902. Epub 2016 Oct 11. J Biomed Mater Res A. 2017. PMID: 27648522 Free PMC article.
References
-
- Geddis AE, Kaushansky K. Immunology: the root of platelet production. Science. 2007;317(5845):1689–1691. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

