Risk assessment model for first-cycle chemotherapy-induced neutropenia in patients with solid tumours
- PMID: 20088918
- PMCID: PMC3082427
- DOI: 10.1111/j.1365-2354.2009.01121.x
Risk assessment model for first-cycle chemotherapy-induced neutropenia in patients with solid tumours
Abstract
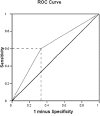
Chemotherapy-induced neutropenia, the major dose-limiting toxicity of chemotherapy, is directly associated with concomitant morbidity, mortality and health-care costs. The use of prophylactic granulocyte colony-stimulating factors may reduce the incidence and duration of chemotherapy-induced neutropenia, and is recommended in high-risk patients. The objective of this study was to develop a model to predict first-cycle chemotherapy-induced neutropenia (defined as neutropenia grade>or=3, with or without body temperature>or=38 degrees C) in patients with solid tumours. A total of 1194 patients [56% women; mean age 58+/-12 years; 94% Eastern Cooperative Oncology Group (ECOG) status<or=1] with solid tumours were included in a multi-centre non-interventional prospective cohort study. A predictive logistic regression model was developed. Several factors were found to influence chemotherapy-induced neutropenia. Higher ECOG status values increased toxicity (ECOG 2 vs. 0, P=0.003; odds ratio 3.12), whereas baseline lymphocyte (P=0.011; odds ratio 0.67) and neutrophil counts (P=0.026; odds ratio 0.90) were inversely related to neutropenia occurrence. Sex and treatment intention also significantly influenced chemotherapy-induced neutropenia (P=0.012). The sensitivity and specificity of the model were 63% and 67% respectively, and the positive and negative predictive values were 17% and 94% respectively. Once validated, this model should be a useful tool for clinical decision making.
Figures
References
-
- Aapro MS, Cameron DA, Pettengell R, Bohlius J, Crawford J, Ellis M, Kearney N, Lyman GH, Tjan-Heijnen VC, Walewski J, Weber DC, Zielinski C. EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphomas and solid tumours. European Journal of Cancer. 2006;42:2433–2453. - PubMed
-
- Aslani A, Smith RC, Allen BJ, Pavlakis N, Levi JA. The predictive value of body protein for chemotherapy-induced toxicity. Cancer. 2000;88:796–803. - PubMed
-
- Blay JY, Chauvin F, Le Cesne A, Anglaret B, Bouhour D, Lasset C, Freyer G, Philip T, Biron P. Early lymphopenia after cytotoxic chemotherapy as a risk factor for febrile neutropenia. Journal of Clinical Oncology. 1996;14:636–643. - PubMed
-
- Bonadonna G, Valagussa P, Moliterni A, Zambetti M, Brambilla C. Adjuvant cyclophosphamide, methotrexate, and fluorouracil in node-positive breast cancer: the results of 20 years of follow-up. The New England Journal of Medicine. 1995;332:901–906. - PubMed
-
- Caggiano V, Weiss RV, Rickert TS, Linde-Zwirble WT. Incidence, cost, and mortality of neutropenia hospitalization associated with chemotherapy. Cancer. 2005;103:1916–1924. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

