Ryanodine receptors contribute to the induction of nociceptive input-evoked long-term potentiation in the rat spinal cord slice
- PMID: 20089138
- PMCID: PMC2826347
- DOI: 10.1186/1744-8069-6-1
Ryanodine receptors contribute to the induction of nociceptive input-evoked long-term potentiation in the rat spinal cord slice
Abstract
Background: Our previous study demonstrated that nitric oxide (NO) contributes to long-term potentiation (LTP) of C-fiber-evoked field potentials by tetanic stimulation of the sciatic nerve in the spinal cord in vivo. Ryanodine receptor (RyR) is a downstream target for NO. The present study further explored the role of RyR in synaptic plasticity of the spinal pain pathway.
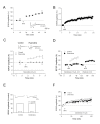
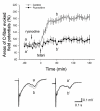
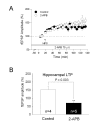
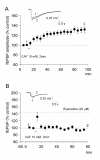
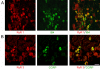
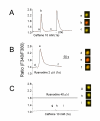
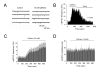
Results: By means of field potential recordings in the adult male rat in vivo, we showed that RyR antagonist reduced LTP of C-fiber-evoked responses in the spinal dorsal horn by tetanic stimulation of the sciatic nerve. Using spinal cord slice preparations and field potential recordings from superficial dorsal horn, high frequency stimulation of Lissauer's tract (LT) stably induced LTP of field excitatory postsynaptic potentials (fEPSPs). Perfusion of RyR antagonists blocked the induction of LT stimulation-evoked spinal LTP, while Ins(1,4,5)P3 receptor (IP(3)R) antagonist had no significant effect on LTP induction. Moreover, activation of RyRs by caffeine without high frequency stimulation induced a long-term potentiation in the presence of bicuculline methiodide and strychnine. Further, in patch-clamp recordings from superficial dorsal horn neurons, activation of RyRs resulted in a large increase in the frequency of miniature EPSCs (mEPSCs). Immunohistochemical study showed that RyRs were expressed in the dorsal root ganglion (DRG) neurons. Likewise, calcium imaging in small DRG neurons illustrated that activation of RyRs elevated [Ca(2+)]i in small DRG neurons.
Conclusions: These data indicate that activation of presynaptic RyRs play a crucial role in the induction of LTP in the spinal pain pathway, probably through enhancement of transmitter release.
Figures

Similar articles
-
Clonidine depresses LTP of C-fiber evoked field potentials in spinal dorsal horn via NO-cGMP pathway.Brain Res. 2006 Nov 6;1118(1):58-65. doi: 10.1016/j.brainres.2006.08.009. Epub 2006 Sep 1. Brain Res. 2006. PMID: 16950233
-
Involvement of ryanodine receptors in tetanic sciatic stimulation-induced long-term potentiation of spinal dorsal horn and persistent pain in rats.J Neurosci Res. 2012 May;90(5):1096-104. doi: 10.1002/jnr.22799. Epub 2012 Feb 8. J Neurosci Res. 2012. PMID: 22315169
-
The effects of Zn2+ on long-term potentiation of C fiber-evoked potentials in the rat spinal dorsal horn.Brain Res Bull. 2001 Dec;56(6):575-9. doi: 10.1016/s0361-9230(01)00728-6. Brain Res Bull. 2001. PMID: 11786244
-
Long-term potentiation at spinal C-fiber synapses: a target for pathological pain.Curr Pharm Des. 2015;21(7):895-905. doi: 10.2174/1381612820666141027115949. Curr Pharm Des. 2015. PMID: 25345608 Review.
-
Role of voltage-gated calcium channels in ascending pain pathways.Brain Res Rev. 2009 Apr;60(1):84-9. doi: 10.1016/j.brainresrev.2008.12.021. Epub 2008 Dec 31. Brain Res Rev. 2009. PMID: 19162069 Free PMC article. Review.
Cited by
-
Electrical stimulation induces calcium-dependent neurite outgrowth and immediate early genes expressions of dorsal root ganglion neurons.Neurochem Res. 2014 Jan;39(1):129-41. doi: 10.1007/s11064-013-1197-7. Epub 2013 Nov 19. Neurochem Res. 2014. PMID: 24248860
-
Ionotropic glutamate receptors and voltage-gated Ca²⁺ channels in long-term potentiation of spinal dorsal horn synapses and pain hypersensitivity.Neural Plast. 2013;2013:654257. doi: 10.1155/2013/654257. Epub 2013 Oct 2. Neural Plast. 2013. PMID: 24224102 Free PMC article. Review.
-
Non-canonical Molecular Targets for Novel Analgesics: Intracellular Calcium and HCN Channels.Curr Neuropharmacol. 2021;19(11):1937-1951. doi: 10.2174/1570159X19666210119153047. Curr Neuropharmacol. 2021. PMID: 33463473 Free PMC article. Review.
-
Involvement of estrogen in rapid pain modulation in the rat spinal cord.Neurochem Res. 2012 Dec;37(12):2697-705. doi: 10.1007/s11064-012-0859-1. Epub 2012 Aug 19. Neurochem Res. 2012. PMID: 22903468
-
Calcium release from intra-axonal endoplasmic reticulum leads to axon degeneration through mitochondrial dysfunction.J Neurosci. 2014 May 21;34(21):7179-89. doi: 10.1523/JNEUROSCI.4784-13.2014. J Neurosci. 2014. PMID: 24849352 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

