Functional consequences of seven novel mutations in the CYP11B1 gene: four mutations associated with nonclassic and three mutations causing classic 11{beta}-hydroxylase deficiency
- PMID: 20089618
- PMCID: PMC2846960
- DOI: 10.1210/jc.2009-0651
Functional consequences of seven novel mutations in the CYP11B1 gene: four mutations associated with nonclassic and three mutations causing classic 11{beta}-hydroxylase deficiency
Abstract
Context: Steroid 11beta-hydroxylase (CYP11B1) deficiency (11OHD) is the second most common form of congenital adrenal hyperplasia (CAH). Cases of nonclassic 11OHD are rare compared with the incidence of nonclassic 21-hydroxylase deficiency.
Objective: The aim of the study was to analyze the functional consequences of seven novel CYP11B1 mutations (p.M88I, p.W116G, p.P159L, p.A165D, p.K254_A259del, p.R366C, p.T401A) found in three patients with classic 11OHD, two patients with nonclassic 11OHD, and three heterozygous carriers for CYP11B1 mutations.
Methods: We conducted functional studies employing a COS7 cell in vitro expression system comparing wild-type (WT) and mutant CYP11B1 activity. Mutants were examined in a computational three-dimensional model of the CYP11B1 protein.
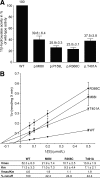
Results: All mutations (p.W116G, p.A165D, p.K254_A259del) found in patients with classic 11OHD have absent or very little 11beta-hydroxylase activity relative to WT. The mutations detected in patients with nonclassic 11OHD showed partial functional impairment, with one patient being homozygous (p.P159L; 25% of WT) and the other patient compound heterozygous for a novel mild p.M88I (40% of WT) and the known severe p.R383Q mutation. The two mutations detected in heterozygous carriers (p.R366C, p.T401A) also reduced CYP11B1 activity by 23 to 37%, respectively.
Conclusion: Functional analysis results allow for the classification of novel CYP11B1 mutations as causative for classic and nonclassic 11OHD, respectively. Four partially inactivating mutations are predicted to result in nonclassic 11OHD. These findings double the number of mild CYP11B1 mutations previously described as associated with mild 11OHD. Our data are important to predict phenotypic expression and provide important information for clinical and genetic counseling in 11OHD.
Figures

Similar articles
-
Characterisation of three novel CYP11B1 mutations in classic and non-classic 11β-hydroxylase deficiency.Eur J Endocrinol. 2014 Apr 10;170(5):697-706. doi: 10.1530/EJE-13-0737. Print 2014 May. Eur J Endocrinol. 2014. PMID: 24536089
-
Characterization of the molecular genetic pathology in patients with 11β-hydroxylase deficiency.Clin Endocrinol (Oxf). 2015 Nov;83(5):629-35. doi: 10.1111/cen.12834. Epub 2015 Jul 14. Clin Endocrinol (Oxf). 2015. PMID: 26053152
-
A diagnosis not to be missed: nonclassic steroid 11β-hydroxylase deficiency presenting with premature adrenarche and hirsutism.J Clin Endocrinol Metab. 2013 Oct;98(10):E1620-5. doi: 10.1210/jc.2013-1306. Epub 2013 Aug 12. J Clin Endocrinol Metab. 2013. PMID: 23940125
-
Non-classical 11β-hydroxylase deficiency caused by compound heterozygous mutations: a case study and literature review.J Ovarian Res. 2018 Sep 17;11(1):82. doi: 10.1186/s13048-018-0450-8. J Ovarian Res. 2018. PMID: 30223866 Free PMC article. Review.
-
Non-classical 11β-hydroxylase deficiency caused by a novel heterozygous mutation: a case report and review of the literature.Endocrine. 2024 Jun;84(3):1193-1205. doi: 10.1007/s12020-024-03746-y. Epub 2024 Feb 27. Endocrine. 2024. PMID: 38411873 Review.
Cited by
-
Getting pregnant with congenital adrenal hyperplasia: Assisted reproduction and pregnancy complications. A systematic review and meta-analysis.Front Endocrinol (Lausanne). 2022 Aug 31;13:982953. doi: 10.3389/fendo.2022.982953. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36120452 Free PMC article.
-
A Novel Mutation in the CYP11B1 Gene Causes Steroid 11β-Hydroxylase Deficient Congenital Adrenal Hyperplasia with Reversible Cardiomyopathy.Int J Endocrinol. 2015;2015:595164. doi: 10.1155/2015/595164. Epub 2015 Jul 22. Int J Endocrinol. 2015. PMID: 26265915 Free PMC article.
-
Clinical perspectives in congenital adrenal hyperplasia due to 11β-hydroxylase deficiency.Endocrine. 2017 Jan;55(1):19-36. doi: 10.1007/s12020-016-1189-x. Epub 2016 Dec 7. Endocrine. 2017. PMID: 27928728 Review.
-
Molecular Recognition in Mitochondrial Cytochromes P450 That Catalyze the Terminal Steps of Corticosteroid Biosynthesis.Biochemistry. 2017 May 2;56(17):2282-2293. doi: 10.1021/acs.biochem.7b00034. Epub 2017 Apr 17. Biochemistry. 2017. PMID: 28355486 Free PMC article.
-
Human cytochrome P450 11B2 produces aldosterone by a processive mechanism due to the lactol form of the intermediate 18-hydroxycorticosterone.J Biol Chem. 2019 Aug 30;294(35):12975-12991. doi: 10.1074/jbc.RA119.009830. Epub 2019 Jul 11. J Biol Chem. 2019. PMID: 31296661 Free PMC article.
References
-
- White PC, Curnow KM, Pascoe L 1994 Disorders of steroid 11 β-hydroxylase isozymes. Endocr Rev 15:421–438 - PubMed
-
- Joehrer K, Geley S, Strasser-Wozak EM, Azziz R, Wollmann HA, Schmitt K, Kofler R, White PC 1997 CYP11B1 mutations causing non-classic adrenal hyperplasia due to 11 β-hydroxylase deficiency. Hum Mol Genet 6:1829–1834 - PubMed
-
- Peters CJ, Nugent T, Perry LA, Davies K, Morel Y, Drake WM, Savage MO, Johnston LB 2007 Cosegregation of a novel homozygous CYP11B1 mutation with the phenotype of non-classical congenital adrenal hyperplasia in a consanguineous family. Horm Res 67:189–193 - PubMed
-
- Speiser PW, White PC 2003 Congenital adrenal hyperplasia. N Engl J Med 349:776–788 - PubMed
-
- Mornet E, Dupont J, Vitek A, White PC 1989 Characterization of two genes encoding human steroid 11 β-hydroxylase (P-450(11) β). J Biol Chem 264:20961–20967 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

