Internalization of coxsackievirus A9 is mediated by {beta}2-microglobulin, dynamin, and Arf6 but not by caveolin-1 or clathrin
- PMID: 20089652
- PMCID: PMC2838097
- DOI: 10.1128/JVI.01340-09
Internalization of coxsackievirus A9 is mediated by {beta}2-microglobulin, dynamin, and Arf6 but not by caveolin-1 or clathrin
Abstract
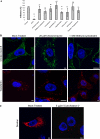
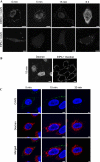
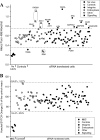
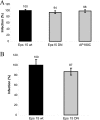
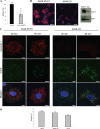
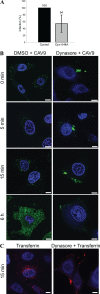
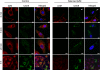
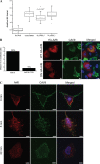
Coxsackievirus A9 (CAV9) is a member of the human enterovirus B species within the Enterovirus genus of the family Picornaviridae. It has been shown to utilize alphaV integrins, particularly alphaVbeta6, as its receptors. The endocytic pathway by which CAV9 enters human cells after the initial attachment to the cell surface has so far been unknown. Here, we present a systematic study concerning the internalization mechanism of CAV9 to A549 human lung carcinoma cells. The small interfering RNA (siRNA) silencing of integrin beta6 subunit inhibited virus proliferation, confirming that alphaVbeta6 mediates the CAV9 infection. However, siRNAs against integrin-linked signaling molecules, such as Src, Fyn, RhoA, phosphatidylinositol 3-kinase, and Akt1, did not reduce CAV9 proliferation, suggesting that the internalization of the virus does not involve integrin-linked signaling events. CAV9 endocytosis was independent of clathrin or caveolin-1 but was restrained by dynasore, an inhibitor of dynamin. The RNA interference silencing of beta2-microglobulin efficiently inhibited virus infection and caused CAV9 to accumulate on the cell surface. Furthermore, CAV9 infection was found to depend on Arf6 as both silencing of this molecule by siRNA and the expression of a dominant negative construct resulted in decreased virus infection. In conclusion, the internalization of CAV9 to A549 cells follows an endocytic pathway that is dependent on integrin alphaVbeta6, beta2-microglobulin, dynamin, and Arf6 but independent of clathrin and caveolin-1.
Figures

References
-
- Benmerah, A., M. Bayrou, N. Cerf-Bensussan, and A. Dautry-Varsat. 1999. Inhibition of clathrin-coated pit assembly by an Eps15 mutant. J. Cell Sci. 112:1303-1311. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

