A specific form of phospho protein phosphatase 2 regulates anaphase-promoting complex/cyclosome association with spindle poles
- PMID: 20089842
- PMCID: PMC2836970
- DOI: 10.1091/mbc.e09-07-0598
A specific form of phospho protein phosphatase 2 regulates anaphase-promoting complex/cyclosome association with spindle poles
Abstract
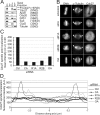
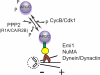
In early mitosis, the END (Emi1/NuMA/Dynein-dynactin) network anchors the anaphase-promoting complex/cyclosome (APC/C) to the mitotic spindle and poles. Spindle anchoring restricts APC/C activity, thereby limiting the destruction of spindle-associated cyclin B and ensuring maintenance of spindle integrity. Emi1 binds directly to hypophosphorylated APC/C, linking the APC/C to the spindle via NuMA. However, whether the phosphorylation state of the APC/C is important for its association with the spindle and what kinases and phosphatases are necessary for regulating this event remain unknown. Here, we describe the regulation of APC/C-mitotic spindle pole association by phosphorylation. We find that only hypophosphorylated APC/C associates with microtubule asters, suggesting that phosphatases are important. Indeed, a specific form of PPP2 (CA/R1A/R2B) binds APC/C, and PPP2 activity is necessary for Cdc27 dephosphorylation. Screening by RNA interference, we find that inactivation of CA, R1A, or R2B leads to delocalization of APC/C from spindle poles, early mitotic spindle defects, a failure to congress chromosomes, and decreased levels of cyclin B on the spindle. Consistently, inhibition of cyclin B/Cdk1 activity increased APC/C binding to microtubules. Thus, cyclin B/Cdk1 and PPP2 regulate the dynamic association of APC/C with spindle poles in early mitosis, a step necessary for proper spindle formation.
Figures




References
-
- Andersen J. S., Wilkinson C. J., Mayor T., Mortensen P., Nigg E. A., Mann M. Proteomic characterization of the human centrosome by protein correlation profiling. Nature. 2003;426:570–574. - PubMed
-
- Ban K. H., Torres J. Z., Miller J. J., Mikhailov A., Nachury M. V., Tung J. J., Rieder C. L., Jackson P. K. The END network couples spindle pole assembly to inhibition of the anaphase-promoting complex/cyclosome in early mitosis. Dev. Cell. 2007;13:29–42. - PubMed
-
- Belmont L. D., Hyman A. A., Sawin K. E., Mitchison T. J. Real-time visualization of cell cycle-dependent changes in microtubule dynamics in cytoplasmic extracts. Cell. 1990;62:579–589. - PubMed
-
- Chen F., et al. Multiple protein phosphatases are required for mitosis in Drosophila. Curr. Biol. 2007;17:293–303. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

