Analysis of the potential role of GluA4 carboxyl-terminus in PDZ interactions
- PMID: 20090852
- PMCID: PMC2806832
- DOI: 10.1371/journal.pone.0008715
Analysis of the potential role of GluA4 carboxyl-terminus in PDZ interactions
Abstract
Background: Specific delivery to synapses of alpha-amino-3-hydroxy-5-methylisoxazole-4-propionate (AMPA) receptors with long-tailed subunits is believed to be a key event in many forms of activity-dependent changes in synaptic strength. GluA1, the best characterized long-tailed AMPA receptor subunit, contains a C-terminal class I PDZ binding motif, which mediates its interaction with scaffold and trafficking proteins, including synapse-associated protein 97 (SAP97). In GluA4, another long-tailed subunit implicated in synaptic plasticity, the PDZ motif is blocked by a single proline residue. This feature is highly conserved in vertebrates, whereas the closest invertebrate homologs of GluA4 have a canonical class I PDZ binding motif. In this work, we have examined the role of GluA4 in PDZ interactions.
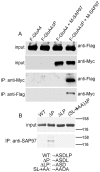
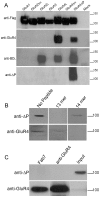
Methodology/principal findings: Deletion of the carboxy-terminal proline residue of recombinant GluA4 conferred avid binding to SAP97 in cultured cells as shown by coimmunoprecipitation, whereas wild-type GluA4 did not associate with SAP97. Native GluA4 and SAP97 coimmunoprecipitated from mouse brain independently of the GluA1 subunit, supporting the possibility of in vivo PDZ interaction. To obtain evidence for or against the exposure of the PDZ motif by carboxyterminal processing of native GluA4 receptors, we generated an antibody reagent specific for proline-deleted GluA4 C-terminus. Immunoprecipitation and mass spectrometric analyses indicated that the carboxyl-terminus of native GluA4 AMPA receptors is intact and that the postulated single-residue cleavage does not occur to any significant extent.
Conclusion/significance: We conclude that native GluA4 receptors are not capable of canonical PDZ interactions and that their association with SAP97 is likely to be indirect.
Conflict of interest statement
Figures


Similar articles
-
Selective binding of synapse-associated protein 97 to GluR-A alpha-amino-5-hydroxy-3-methyl-4-isoxazole propionate receptor subunit is determined by a novel sequence motif.J Biol Chem. 2002 Aug 30;277(35):31484-90. doi: 10.1074/jbc.M204354200. Epub 2002 Jun 17. J Biol Chem. 2002. PMID: 12070168
-
Sequential delivery of synaptic GluA1- and GluA4-containing AMPA receptors (AMPARs) by SAP97 anchored protein complexes in classical conditioning.J Biol Chem. 2014 Apr 11;289(15):10540-10550. doi: 10.1074/jbc.M113.535179. Epub 2014 Feb 24. J Biol Chem. 2014. PMID: 24567325 Free PMC article.
-
SAP97 is associated with the alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptor GluR1 subunit.J Biol Chem. 1998 Jul 31;273(31):19518-24. doi: 10.1074/jbc.273.31.19518. J Biol Chem. 1998. PMID: 9677374
-
Interactions between AMPA receptors and intracellular proteins.Neuropharmacology. 2000 Apr 3;39(6):919-30. doi: 10.1016/s0028-3908(99)00171-9. Neuropharmacology. 2000. PMID: 10727702 Review.
-
The anchoring protein SAP97 influences the trafficking and localisation of multiple membrane channels.Biochim Biophys Acta. 2014 Feb;1838(2):589-94. doi: 10.1016/j.bbamem.2013.03.015. Epub 2013 Mar 25. Biochim Biophys Acta. 2014. PMID: 23535319 Review.
Cited by
-
Bi-factor analysis based on noise-reduction (BIFANR): a new algorithm for detecting coevolving amino acid sites in proteins.PLoS One. 2013 Nov 20;8(11):e79764. doi: 10.1371/journal.pone.0079764. eCollection 2013. PLoS One. 2013. PMID: 24278175 Free PMC article.
-
Splicing and editing of ionotropic glutamate receptors: a comprehensive analysis based on human RNA-Seq data.Cell Mol Life Sci. 2021 Jul;78(14):5605-5630. doi: 10.1007/s00018-021-03865-z. Epub 2021 Jun 8. Cell Mol Life Sci. 2021. PMID: 34100982 Free PMC article.
-
Structural position correlation analysis (SPCA) for protein family.PLoS One. 2011;6(12):e28206. doi: 10.1371/journal.pone.0028206. Epub 2011 Dec 5. PLoS One. 2011. PMID: 22163002 Free PMC article.
-
The N-terminal domain modulates α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor desensitization.J Biol Chem. 2014 May 9;289(19):13197-205. doi: 10.1074/jbc.M113.526301. Epub 2014 Mar 20. J Biol Chem. 2014. PMID: 24652293 Free PMC article.
-
Developmental switch in the kinase dependency of long-term potentiation depends on expression of GluA4 subunit-containing AMPA receptors.Proc Natl Acad Sci U S A. 2014 Mar 18;111(11):4321-6. doi: 10.1073/pnas.1315769111. Epub 2014 Mar 5. Proc Natl Acad Sci U S A. 2014. PMID: 24599589 Free PMC article.
References
-
- Bredt DS, Nicoll RA. AMPA receptor trafficking at excitatory synapses. Neuron. 2003;40:361–379. - PubMed
-
- Shepherd JD, Huganir RL. The cell biology of synaptic plasticity: AMPA receptor trafficking. Annu Rev Cell Dev Biol. 2007;23:613–643. - PubMed
-
- Shi S, Hayashi Y, Esteban JA, Malinow R. Subunit-specific rules governing AMPA receptor trafficking to synapses in hippocampal pyramidal neurons. Cell. 2001;105:331–343. - PubMed
-
- Passafaro M, Piech V, Sheng M. Subunit-specific temporal and spatial patterns of AMPA receptor exocytosis in hippocampal neurons. Nat Neurosci. 2001;4:917–926. - PubMed
-
- Zhu JJ, Esteban JA, Hayashi Y, Malinow R. Postnatal synaptic potentiation: Delivery of GluR4-containing AMPA receptors by spontaneous activity. Nat Neurosci. 2000;3:1098–1106. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

