Initial characterization of an immunotoxin constructed from domains II and III of cholera exotoxin
- PMID: 20091030
- PMCID: PMC7394282
- DOI: 10.1007/s00262-009-0794-4
Initial characterization of an immunotoxin constructed from domains II and III of cholera exotoxin
Abstract
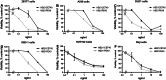
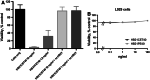
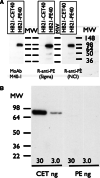
Immunotoxins are antibody-toxin fusion proteins under development as cancer therapeutics. In early clinical trials, immunotoxins constructed with domains II and III of Pseudomonas exotoxin (termed PE38), have produced a high rate of complete remissions in Hairy Cell Leukemia and objective responses in other malignancies. Cholera exotoxin (also known as cholix toxin) has a very similar three-dimensional structure to Pseudomonas exotoxin (PE) and when domains II and III of each are compared at the primary sequence level, they are 36% identical and 50% similar. Here we report on the construction and activity of an immunotoxin made with domains II and III of cholera exotoxin (here termed CET40). In cell viability assays, the CET40 immunotoxin was equipotent to tenfold less active compared to a PE-based immunotoxin made with the same single-chain Fv. A major limitation of toxin-based immunotoxins is the development of neutralizing antibodies to the toxin portion of the immunotoxin. Because of structure and sequence similarities, we evaluated a CET40 immunotoxin for the presence of PE-related epitopes. In western blots, three-of-three anti-PE antibody preparations failed to react with the CET40 immunotoxin. More importantly, in neutralization studies neither these antibodies nor those from patients with neutralizing titers to PE38, neutralized the CET40-immunotoxin. We propose that the use of modular components such as antibody Fvs and toxin domains will allow a greater flexibility in how these agents are designed and deployed including the sequential administration of a second immunotoxin after patients have developed neutralizing antibodies to the first.
Figures

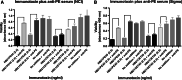
Similar articles
-
Influence of relative binding affinity on efficacy in a panel of anti-CD3 scFv immunotoxins.Mol Immunol. 2001 Sep;38(5):397-408. doi: 10.1016/s0161-5890(01)00070-0. Mol Immunol. 2001. PMID: 11684296
-
Identification of epitopes on a mutant form of Pseudomonas exotoxin using serum from humans treated with Pseudomonas exotoxin containing immunotoxins.Eur J Immunol. 1997 Jun;27(6):1459-68. doi: 10.1002/eji.1830270624. Eur J Immunol. 1997. PMID: 9209499 Clinical Trial.
-
Primate antibody response to immunotoxin: serological and computer-aided analysis of epitopes on a truncated form of Pseudomonas exotoxin.Infect Immun. 1994 Nov;62(11):5055-65. doi: 10.1128/iai.62.11.5055-5065.1994. Infect Immun. 1994. PMID: 7927788 Free PMC article.
-
Immunogenicity of Immunotoxins Containing Pseudomonas Exotoxin A: Causes, Consequences, and Mitigation.Front Immunol. 2020 Jun 26;11:1261. doi: 10.3389/fimmu.2020.01261. eCollection 2020. Front Immunol. 2020. PMID: 32695104 Free PMC article. Review.
-
Immunogenicity of therapeutic recombinant immunotoxins.Immunol Rev. 2016 Mar;270(1):152-64. doi: 10.1111/imr.12390. Immunol Rev. 2016. PMID: 26864110 Free PMC article. Review.
Cited by
-
The Father, Son and Cholix Toxin: The Third Member of the DT Group Mono-ADP-Ribosyltransferase Toxin Family.Toxins (Basel). 2015 Jul 24;7(8):2757-72. doi: 10.3390/toxins7082757. Toxins (Basel). 2015. PMID: 26213968 Free PMC article. Review.
-
Pseudomonas Exotoxin-Based Immunotoxins: Over Three Decades of Efforts on Targeting Cancer Cells With the Toxin.Front Oncol. 2021 Dec 16;11:781800. doi: 10.3389/fonc.2021.781800. eCollection 2021. Front Oncol. 2021. PMID: 34976821 Free PMC article. Review.
-
Synthesis of a covalent epirubicin-(C(3)-amide)-anti-HER2/neu immunochemotherapeutic utilizing a UV-photoactivated anthracycline intermediate.Cancer Biother Radiopharm. 2012 Feb;27(1):41-55. doi: 10.1089/cbr.2011.1097. Epub 2011 Dec 22. Cancer Biother Radiopharm. 2012. PMID: 22191802 Free PMC article.
-
Antibody-Based Immunotoxins for Colorectal Cancer Therapy.Biomedicines. 2021 Nov 19;9(11):1729. doi: 10.3390/biomedicines9111729. Biomedicines. 2021. PMID: 34829955 Free PMC article. Review.
-
A guide to taming a toxin--recombinant immunotoxins constructed from Pseudomonas exotoxin A for the treatment of cancer.FEBS J. 2011 Dec;278(23):4683-700. doi: 10.1111/j.1742-4658.2011.08182.x. Epub 2011 Jun 2. FEBS J. 2011. PMID: 21585657 Free PMC article. Review.
References
-
- Heimann DM, Weiner LM. Monoclonal antibodies in therapy of solid tumors. Surg Oncol Clin N Am. 2007;16:775–792. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

