Cell-specific and lamin-dependent targeting of novel transmembrane proteins in the nuclear envelope
- PMID: 20091084
- PMCID: PMC2839517
- DOI: 10.1007/s00018-010-0257-2
Cell-specific and lamin-dependent targeting of novel transmembrane proteins in the nuclear envelope
Abstract
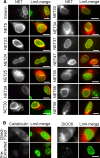
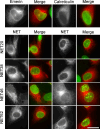
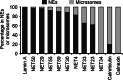
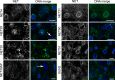
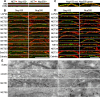
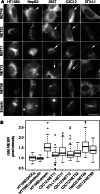
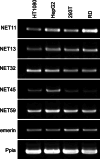
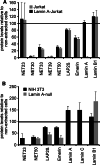
Nuclear envelope complexity is expanding with respect to identification of protein components. Here we test the validity of proteomics results that identified 67 novel predicted nuclear envelope transmembrane proteins (NETs) from liver by directly comparing 30 as tagged fusions using targeting assays. This confirmed 21 as NETs, but 4 only targeted in certain cell types, underscoring the complexity of interactions that tether NETs to the nuclear envelope. Four NETs accumulated at the nuclear rim in normal fibroblasts but not in fibroblasts lacking lamin A, suggesting involvement of lamin A in tethering them in the nucleus. However, intriguingly, for the NETs tested alternative mechanisms for nuclear envelope retention could be found in Jurkat cells that normally lack lamin A. This study expands by a factor of three the number of liver NETs analyzed, bringing the total confirmed to 31, and shows that several have multiple mechanisms for nuclear envelope retention.
Figures

Similar articles
-
A role for β-dystroglycan in the organization and structure of the nucleus in myoblasts.Biochim Biophys Acta. 2013 Mar;1833(3):698-711. doi: 10.1016/j.bbamcr.2012.11.019. Epub 2012 Dec 4. Biochim Biophys Acta. 2013. PMID: 23220011
-
Nuclear envelope localization of LEMD2 is developmentally dynamic and lamin A/C dependent yet insufficient for heterochromatin tethering.Differentiation. 2017 Mar-Apr;94:58-70. doi: 10.1016/j.diff.2016.12.002. Epub 2017 Jan 2. Differentiation. 2017. PMID: 28056360
-
Nuclear envelope transmembrane proteins (NETs) that are up-regulated during myogenesis.BMC Cell Biol. 2006 Oct 24;7:38. doi: 10.1186/1471-2121-7-38. BMC Cell Biol. 2006. PMID: 17062158 Free PMC article.
-
Identification of novel integral membrane proteins of the nuclear envelope with potential disease links using subtractive proteomics.Novartis Found Symp. 2005;264:63-76; discussion 76-80, 227-30. Novartis Found Symp. 2005. PMID: 15773748 Review.
-
Dysfunction of lamin A triggers a DNA damage response and cellular senescence.DNA Repair (Amst). 2006 Feb 3;5(2):286-9. doi: 10.1016/j.dnarep.2005.10.007. Epub 2005 Dec 15. DNA Repair (Amst). 2006. PMID: 16344005 Review.
Cited by
-
An atlas of lamina-associated chromatin across twelve human cell types reveals an intermediate chromatin subtype.Genome Biol. 2023 Jan 23;24(1):16. doi: 10.1186/s13059-023-02849-5. Genome Biol. 2023. PMID: 36691074 Free PMC article.
-
Transmembrane proteins with unknown function (TMEMs) as ion channels: electrophysiological properties, structure, and pathophysiological roles.Exp Mol Med. 2024 Apr;56(4):850-860. doi: 10.1038/s12276-024-01206-1. Epub 2024 Apr 1. Exp Mol Med. 2024. PMID: 38556553 Free PMC article. Review.
-
STING1 in Different Organelles: Location Dictates Function.Front Immunol. 2022 Mar 17;13:842489. doi: 10.3389/fimmu.2022.842489. eCollection 2022. Front Immunol. 2022. PMID: 35371032 Free PMC article. Review.
-
Lamina-associated domains: peripheral matters and internal affairs.Genome Biol. 2020 Apr 2;21(1):85. doi: 10.1186/s13059-020-02003-5. Genome Biol. 2020. PMID: 32241294 Free PMC article. Review.
-
Unique and shared functions of nuclear lamina LEM domain proteins in Drosophila.Genetics. 2014 Jun;197(2):653-65. doi: 10.1534/genetics.114.162941. Epub 2014 Apr 3. Genetics. 2014. PMID: 24700158 Free PMC article.
References
-
- Bonne G, Di Barletta MR, Varnous S, Becane HM, Hammouda EH, Merlini L, Muntoni F, Greenberg CR, Gary F, Urtizberea JA, Duboc D, Fardeau M, Toniolo D, Schwartz K. Mutations in the gene encoding lamin A/C cause autosomal dominant Emery-Dreifuss muscular dystrophy. Nat Genet. 1999;21:285–288. doi: 10.1038/6799. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

