PfPI3K, a phosphatidylinositol-3 kinase from Plasmodium falciparum, is exported to the host erythrocyte and is involved in hemoglobin trafficking
- PMID: 20093402
- PMCID: PMC2918364
- DOI: 10.1182/blood-2009-08-238972
PfPI3K, a phosphatidylinositol-3 kinase from Plasmodium falciparum, is exported to the host erythrocyte and is involved in hemoglobin trafficking
Abstract
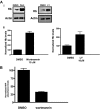
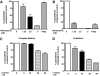
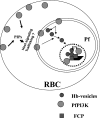
Polyphosphorylated phosphoinositides (PIPs) are potent second messengers, which trigger a wide variety of signaling and trafficking events in most eukaryotic cells. However, the role and metabolism of PIPs in malaria parasite Plasmodium have remained largely unexplored. Our present studies suggest that PfPI3K, a novel phosphatidylinositol-3-kinase (PI3K) in Plasmodium falciparum, is exported to the host erythrocyte by the parasite in an active form. PfPI3K is a versatile enzyme as it can generate various 3'-phosphorylated PIPs. In the parasite, PfPI3K was localized in vesicular compartments near the membrane and in its food vacuole. PI3K inhibitors wortmannin and LY294002 were effective against PfPI3K and were used to study PfPI3K function. We found that PfPI3K is involved in endocytosis from the host and trafficking of hemoglobin in the parasite. The inhibition of PfPI3K resulted in entrapment of hemoglobin in vesicles in the parasite cytoplasm, which prevented its transport to the food vacuole, the site of hemoglobin catabolism. As a result, hemoglobin digestion, which is a source of amino acids necessary for parasite growth, was attenuated and caused the inhibition of parasite growth.
Figures
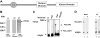

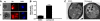
Similar articles
-
Plasmodium falciparum selectively degrades α-spectrin of infected erythrocytes after invasion.mBio. 2024 Apr 10;15(4):e0351023. doi: 10.1128/mbio.03510-23. Epub 2024 Mar 12. mBio. 2024. PMID: 38470053 Free PMC article.
-
A Phosphoinositide-Binding Protein Acts in the Trafficking Pathway of Hemoglobin in the Malaria Parasite Plasmodium falciparum.mBio. 2022 Feb 22;13(1):e0323921. doi: 10.1128/mbio.03239-21. Epub 2022 Jan 18. mBio. 2022. PMID: 35038916 Free PMC article.
-
Phosphatidylinositol 3-phosphate, an essential lipid in Plasmodium, localizes to the food vacuole membrane and the apicoplast.Eukaryot Cell. 2010 Oct;9(10):1519-30. doi: 10.1128/EC.00124-10. Epub 2010 Aug 13. Eukaryot Cell. 2010. PMID: 20709789 Free PMC article.
-
Vesicle-mediated trafficking of parasite proteins to the host cell cytosol and erythrocyte surface membrane in Plasmodium falciparum infected erythrocytes.Int J Parasitol. 2001 Oct;31(12):1381-91. doi: 10.1016/s0020-7519(01)00256-9. Int J Parasitol. 2001. PMID: 11566305 Review.
-
Targeting malaria parasite proteins to the erythrocyte.Trends Parasitol. 2005 Sep;21(9):399-402. doi: 10.1016/j.pt.2005.07.006. Trends Parasitol. 2005. PMID: 16046185 Review.
Cited by
-
Plasmodium falciparum resistance to ACTs: Emergence, mechanisms, and outlook.Int J Parasitol Drugs Drug Resist. 2021 Aug;16:102-118. doi: 10.1016/j.ijpddr.2021.05.007. Epub 2021 May 26. Int J Parasitol Drugs Drug Resist. 2021. PMID: 34090067 Free PMC article.
-
Ingested human insulin inhibits the mosquito NF-κB-dependent immune response to Plasmodium falciparum.Infect Immun. 2012 Jun;80(6):2141-9. doi: 10.1128/IAI.00024-12. Epub 2012 Apr 2. Infect Immun. 2012. PMID: 22473605 Free PMC article.
-
Taste for blood: hemoglobin as a nutrient source for pathogens.PLoS Pathog. 2012;8(3):e1002535. doi: 10.1371/journal.ppat.1002535. Epub 2012 Mar 8. PLoS Pathog. 2012. PMID: 22412370 Free PMC article. No abstract available.
-
Lipid kinases are essential for apicoplast homeostasis in Toxoplasma gondii.Cell Microbiol. 2015 Apr;17(4):559-78. doi: 10.1111/cmi.12383. Epub 2014 Nov 22. Cell Microbiol. 2015. PMID: 25329540 Free PMC article.
-
A VPS15-like kinase regulates apicoplast biogenesis and autophagy by promoting PI3P generation in Toxoplasma gondii.PLoS Pathog. 2022 Nov 1;18(11):e1010922. doi: 10.1371/journal.ppat.1010922. eCollection 2022 Nov. PLoS Pathog. 2022. PMID: 36318587 Free PMC article.
References
-
- Goodyer ID, Pouvelle B, Schneider TG, Trelka DP, Taraschi TF. Characterization of macromolecular transport pathways in malaria-infected erythrocytes. Mol Biochem Parasitol. 1997;87(1):13–28. - PubMed
-
- Francis SE, Sullivan DJ, Jr, Goldberg DE. Hemoglobin metabolism in the malaria parasite Plasmodium falciparum. Annu Rev Microbiol. 1997;51:97–123. - PubMed
-
- Vanhaesebroeck B, Leevers SJ, Ahmadi K, et al. Synthesis and function of 3-phosphorylated inositol lipids. Annu Rev Biochem. 2001;70:535–602. - PubMed
-
- Lemmon MA. Phosphoinositide recognition domains. Traffic. 2003;4(4):201–213. - PubMed
-
- Schu PV, Takegawa K, Fry MJ, et al. Phosphatidylinositol 3-kinase encoded by yeast VPS34 gene essential for protein sorting. Science. 1993;260(5104):88–91. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

