Critical biophysical properties in the Pseudomonas aeruginosa efflux gene regulator MexR are targeted by mutations conferring multidrug resistance
- PMID: 20095047
- PMCID: PMC2867009
- DOI: 10.1002/pro.343
Critical biophysical properties in the Pseudomonas aeruginosa efflux gene regulator MexR are targeted by mutations conferring multidrug resistance
Abstract
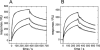
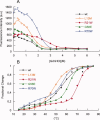
The self-assembling MexA-MexB-OprM efflux pump system, encoded by the mexO operon, contributes to facile resistance of Pseudomonas aeruginosa by actively extruding multiple antimicrobials. MexR negatively regulates the mexO operon, comprising two adjacent MexR binding sites, and is as such highly targeted by mutations that confer multidrug resistance (MDR). To understand how MDR mutations impair MexR function, we studied MexR-wt as well as a selected set of MDR single mutants distant from the proposed DNA-binding helix. Although DNA affinity and MexA-MexB-OprM repression were both drastically impaired in the selected MexR-MDR mutants, MexR-wt bound its two binding sites in the mexO with high affinity as a dimer. In the MexR-MDR mutants, secondary structure content and oligomerization properties were very similar to MexR-wt despite their lack of DNA binding. Despite this, the MexR-MDR mutants showed highly varying stabilities compared with MexR-wt, suggesting disturbed critical interdomain contacts, because mutations in the DNA-binding domains affected the stability of the dimer region and vice versa. Furthermore, significant ANS binding to MexR-wt in both free and DNA-bound states, together with increased ANS binding in all studied mutants, suggest that a hydrophobic cavity in the dimer region already shown to be involved in regulatory binding is enlarged by MDR mutations. Taken together, we propose that the biophysical MexR properties that are targeted by MDR mutations-stability, domain interactions, and internal hydrophobic surfaces-are also critical for the regulation of MexR DNA binding.
Figures
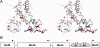

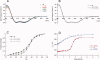
References
-
- Alekshun MN, Levy SB. Molecular mechanisms of antibacterial multidrug resistance. Cell. 2007;128:1037–1050. - PubMed
-
- Gajiwala KS, Burley SK. Winged helix proteins. Curr Opin Struct Biol. 2000;10:110–116. - PubMed
-
- Wilkinson SP, Grove A. Ligand-responsive transcriptional regulation by members of the MarR family of winged helix proteins. Curr Issues Mol Biol. 2006;8:51–62. - PubMed
-
- Poole K. Efflux-mediated antimicrobial resistance. J Antimicrob Chemother. 2005;56:20–51. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

