Molecular dynamics simulations of nonpolarizable inorganic salt solution interfaces: NaCl, NaBr, and NaI in transferable intermolecular potential 4-point with charge dependent polarizability (TIP4P-QDP) water
- PMID: 20095700
- PMCID: PMC2821151
- DOI: 10.1063/1.3269673
Molecular dynamics simulations of nonpolarizable inorganic salt solution interfaces: NaCl, NaBr, and NaI in transferable intermolecular potential 4-point with charge dependent polarizability (TIP4P-QDP) water
Abstract
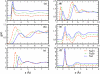
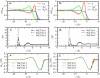
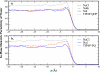
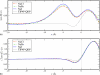
We present molecular dynamics simulations of the liquid-vapor interface of 1M salt solutions of nonpolarizable NaCl, NaBr, and NaI in polarizable transferable intermolecular potential 4-point with charge dependent polarizability water [B. A. Bauer et al., J. Chem. Theory Comput. 5, 359 (2009)]; this water model accommodates increased solvent polarizability (relative to the condensed phase) in the interfacial and vapor regions. We employ fixed-charge ion models developed in conjunction with the TIP4P-QDP water model to reproduce ab initio ion-water binding energies and ion-water distances for isolated ion-water pairs. The transferability of these ion models to the condensed phase was validated with hydration free energies computed using thermodynamic integration (TI) and appropriate energy corrections. Density profiles of Cl(-), Br(-), and I(-) exhibit charge layering in the interfacial region; anions and cation interfacial probabilities show marked localization, with the anions penetrating further toward the vapor than the cations. Importantly, in none of the cases studied do anions favor the outermost regions of the interface; there is always an aqueous region between the anions and vapor phase. Observed interfacial charge layering is independent of the strength of anion-cation interactions as manifest in anion-cation contact ion pair peaks and solvent separated ion pair peaks; by artificially modulating the strength of anion-cation interactions (independent of their interactions with solvent), we find little dependence on charge layering particularly for the larger iodide anion. The present results reiterate the widely held view of the importance of solvent and ion polarizability in mediating specific anion surface segregation effects. Moreover, due to the higher parametrized polarizability of the TIP4P-QDP condensed phase {1.31 A(3) for TIP4P-QDP versus 1.1 A(3) (TIP4P-FQ) and 0.87 A(3) (POL3) [Ponder and Case, Adv. Protein Chem. 66, 27 (2003)]} based on ab initio calculations of the condensed-phase polarizability reduction in liquid water, the present simulations highlight the role of water polarizability in inducing water molecular dipole moments parallel to the interface normal (and within the interfacial region) so as to favorably oppose the macrodipole generated by the separation of anion and cation charge. Since the TIP4P-QDP water polarizability approaches that of the experimental vapor phase value for water, the present results suggest a fundamental role of solvent polarizability in accommodating the large spatial dipole generated by the separation of ion charges. The present results draw further attention to the question of what exact value of condensed phase water polarizability to incorporate in classical polarizable water force fields.
Figures

Similar articles
-
Incorporating Phase-Dependent Polarizability in Non-Additive Electrostatic Models for Molecular Dynamics Simulations of the Aqueous Liquid-Vapor Interface.J Chem Theory Comput. 2009 Feb 10;5(2):359-373. doi: 10.1021/ct800320f. Epub 2009 Jan 8. J Chem Theory Comput. 2009. PMID: 23133341 Free PMC article.
-
Electrostatic properties of aqueous salt solution interfaces: a comparison of polarizable and nonpolarizable ion models.J Phys Chem B. 2008 Sep 18;112(37):11679-93. doi: 10.1021/jp8038835. Epub 2008 Aug 20. J Phys Chem B. 2008. PMID: 18712908 Free PMC article.
-
Properties of water along the liquid-vapor coexistence curve via molecular dynamics simulations using the polarizable TIP4P-QDP-LJ water model.J Chem Phys. 2009 Aug 28;131(8):084709. doi: 10.1063/1.3200869. J Chem Phys. 2009. PMID: 19725623 Free PMC article.
-
An extensive assessment of the performance of pairwise and many-body interaction potentials in reproducing ab initio benchmark binding energies for water clusters n = 2-25.Phys Chem Chem Phys. 2023 Mar 8;25(10):7120-7143. doi: 10.1039/d2cp03241d. Phys Chem Chem Phys. 2023. PMID: 36853239 Review.
-
Ions at hydrophobic interfaces.J Phys Condens Matter. 2014 May 21;26(20):203101. doi: 10.1088/0953-8984/26/20/203101. Epub 2014 Apr 25. J Phys Condens Matter. 2014. PMID: 24769502 Review.
Cited by
-
Influence of Surfactants with Differently Charged Headgroups on the Surface Propensity of Bromide.J Phys Chem A. 2025 Apr 3;129(13):3085-3097. doi: 10.1021/acs.jpca.4c07539. Epub 2025 Mar 21. J Phys Chem A. 2025. PMID: 40118072 Free PMC article.
-
Solvation Structure and Energetics of Single Ions at the Aqueous Liquid-Vapor Interface.Chem Phys Lett. 2012 Feb 27;527:22-26. doi: 10.1016/j.cplett.2011.12.061. Chem Phys Lett. 2012. PMID: 23136448 Free PMC article.
-
Real-time structural dynamics of the ultrafast solvation process around photo-excited aqueous halides.Chem Sci. 2024 Jun 12;15(29):11391-11401. doi: 10.1039/d4sc01912a. eCollection 2024 Jul 24. Chem Sci. 2024. PMID: 39055005 Free PMC article.
-
Spherical monovalent ions at aqueous liquid-vapor interfaces: interfacial stability and induced interface fluctuations.J Phys Chem B. 2013 Oct 3;117(39):11732-42. doi: 10.1021/jp406001b. Epub 2013 Sep 16. J Phys Chem B. 2013. PMID: 24032752 Free PMC article.
References
-
- Piazza R. J., J. Cryst. Growth JCRGAE 196, 415 (1999).10.1016/S0022-0248(98)00867-7 - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

