Modulation of delta opioid agonist-induced antinociception by repeated morphine pretreatment in rhesus monkeys
- PMID: 20096291
- PMCID: PMC2913466
- DOI: 10.1016/j.lfs.2010.01.006
Modulation of delta opioid agonist-induced antinociception by repeated morphine pretreatment in rhesus monkeys
Abstract
Aims: Repeated treatment with morphine increases antinociceptive effects of delta opioid agonists in rodents by a mechanism that may involve increased cell-surface expression of delta receptors. The present study evaluated effects of repeated morphine treatment on behavioral effects of the delta agonist SNC80 and the mu agonist fentanyl in rhesus monkeys.
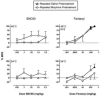
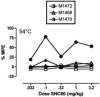
Main methods: In an assay of schedule-controlled responding, three monkeys responded for food reinforcement under a fixed-ratio 30 schedule. In an assay of thermal nociception, tail-withdrawal latencies were evaluated in three monkeys using thermal stimulus intensities of 48 and 54 degrees C. In both assays, the effects of SNC80 (0.032-3.2mg/kg) and fentanyl (0.001-0.056 mg/kg) were evaluated after repeated treatment with saline or a regimen of morphine doses modeled on the regimen that enhanced delta agonist antinociception and apparent delta receptor availability in previous rodent studies.
Key findings: Both SNC80 and fentanyl dose-dependently decreased rates of schedule-controlled responding, and repeated morphine treatment did not significantly alter these effects. In the assay of thermal nociception, SNC80 had little effect on tail-withdrawal latencies from water heated to 48 or 54 degrees C, whereas fentanyl increased tail-withdrawal latencies at both temperatures. Repeated morphine tended to increase the antinociceptive effects of SNC80 and to decrease the antinociceptive effects of fentanyl, but these effects of repeated morphine were small and were significant only at the higher stimulus intensity (54 degrees C).
Significance: These results provide limited support for the proposition that prior stimulation of mu receptors selectively increases the antinociceptive effects of delta agonists in rhesus monkeys.
Copyright (c) 2010 Elsevier Inc. All rights reserved.
Figures



References
-
- Brandt MR, Furness MS, Mello NK, Rice KC, Negus SS. Antinociceptive effects of delta-opioid agonists in Rhesus monkeys: effects on chemically induced thermal hypersensitivity. Journal of Pharmacology and Experimental Therapeutics. 2001;296(3):939–946. - PubMed
-
- Butelman ER, Negus SS, Gatch MB, Chang KJ, Woods JH. BW373U86, a delta-opioid receptor agonist, reverses bradykinin-induced thermal allodynia in rhesus monkeys. European Journal of Pharmacology. 1995;277(2-3):285–287. - PubMed
-
- Cahill CM, Holdridge SV, Morinville A. Trafficking of delta-opioid receptors and other G-protein-coupled receptors: implications for pain and analgesia. Trends in Pharmacological Sciences. 2007;28(1):23–31. - PubMed
-
- Cahill CM, Morinville A, Hoffert C, O’Donnell D, Beaudet A. Up-regulation and trafficking of delta opioid receptor in a model of chronic inflammation: implications for pain control. Pain. 2003;101(1-2):199–208. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

