Distribution of small integrin-binding ligand, N-linked glycoproteins (SIBLING) in the condylar cartilage of rat mandible
- PMID: 20097540
- PMCID: PMC2834875
- DOI: 10.1016/j.ijom.2009.12.017
Distribution of small integrin-binding ligand, N-linked glycoproteins (SIBLING) in the condylar cartilage of rat mandible
Abstract
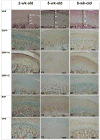
The Small Integrin-Binding LIgand, N-linked Glycoprotein (SIBLING) family is one category of non-collagenous proteins closely related to osteogenesis. In this study, the authors systematically evaluated the presence and distribution of four SIBLING family members, dentin sialophosphoprotein (DSPP), dentin matrix protein 1 (DMP1), bone sialoprotein (BSP) and osteopontin (OPN), in rat mandibular condylar cartilage using protein chemistry and immunohistochemistry. For protein chemistry, SIBLING proteins in the dissected condylar cartilage were extracted with 4M guanidium-HCl, separated by ion-exchange chromatography, and analyzed by Western immunoblotting. Immunohistochemistry was employed to assess the distribution of these four SIBLING proteins in the condylar cartilage of 2-, 5- and 8-week-old rats. Results from both approaches showed that all four members are expressed in the condylar cartilage. DSPP, unlike that observed in dentin and bone, exists as a full-length form (uncleaved) in the condylar cartilage. The NH(2)-terminal fragment of DMP1 is mainly detected in the matrix of the cartilage while the COOH-terminal fragment is primarily localized in the nuclei of cells in the chondroblastic and hypertrophic layers. The data obtained in this investigation provide clues about the potential roles of these SIBLING proteins in chondrogenesis.
Figures



Similar articles
-
Distribution of small integrin-binding ligand, N-linked glycoproteins (SIBLING) in the articular cartilage of the rat femoral head.J Histochem Cytochem. 2010 Nov;58(11):1033-43. doi: 10.1369/jhc.2010.956771. Epub 2010 Aug 2. J Histochem Cytochem. 2010. PMID: 20679519 Free PMC article.
-
Distribution of SIBLING proteins in the organic and inorganic phases of rat dentin and bone.Eur J Oral Sci. 2008 Apr;116(2):104-12. doi: 10.1111/j.1600-0722.2008.00522.x. Eur J Oral Sci. 2008. PMID: 18353003 Free PMC article.
-
Expression and distribution of SIBLING proteins in the predentin/dentin and mandible of hyp mice.Oral Dis. 2010 Jul;16(5):453-64. doi: 10.1111/j.1601-0825.2010.01656.x. Epub 2010 Mar 9. Oral Dis. 2010. PMID: 20233318 Free PMC article.
-
Post-translational modifications of sibling proteins and their roles in osteogenesis and dentinogenesis.Crit Rev Oral Biol Med. 2004 Jun 4;15(3):126-36. doi: 10.1177/154411130401500302. Crit Rev Oral Biol Med. 2004. PMID: 15187031 Review.
-
[SIBLING proteins: molecular tools for tumor progression and angiogenesis].Med Sci (Paris). 2013 Nov;29(11):1018-25. doi: 10.1051/medsci/20132911019. Epub 2013 Nov 20. Med Sci (Paris). 2013. PMID: 24280506 Review. French.
Cited by
-
Glycosylation of Dentin Matrix Protein 1 is critical for osteogenesis.Sci Rep. 2015 Dec 4;5:17518. doi: 10.1038/srep17518. Sci Rep. 2015. PMID: 26634432 Free PMC article.
-
Long-term haplodeficency of DSPP causes temporomandibular joint osteoarthritis in mice.BMC Oral Health. 2024 May 14;24(1):569. doi: 10.1186/s12903-024-04320-8. BMC Oral Health. 2024. PMID: 38745274 Free PMC article.
-
iTRAQ-based quantitative proteomic analysis on differentially expressed proteins of rat mandibular condylar cartilage induced by reducing dietary loading.Front Med. 2017 Mar;11(1):97-109. doi: 10.1007/s11684-016-0496-1. Epub 2017 Mar 2. Front Med. 2017. PMID: 28116632
-
The role of Dentin Sialophosphoprotein (DSPP) in craniofacial development.J Oral Biol Craniofac Res. 2022 Sep-Oct;12(5):673-678. doi: 10.1016/j.jobcr.2022.08.010. Epub 2022 Aug 23. J Oral Biol Craniofac Res. 2022. PMID: 36062256 Free PMC article.
-
Role of the NH2 -terminal fragment of dentin sialophosphoprotein in dentinogenesis.Eur J Oral Sci. 2013 Apr;121(2):76-85. doi: 10.1111/eos.12020. Epub 2013 Feb 7. Eur J Oral Sci. 2013. PMID: 23489896 Free PMC article.
References
-
- Baba O, Qin C, Brunn JC, Jones JE, Wygant JN, McIntyre BW, Butler WT. Detection of dentin sialoprotein in rat periodontium. Eur J Oral Sci. 2004;112:163–170. - PubMed
-
- Baba O, Qin C, Brunn JC, Wygant JN, McIntyre BW, Butler WT. Colocalization of dentin matrix protein 1 and dentin sialoprotein at late stages of rat molar development. Matrix Biol. 2004;23:371–379. - PubMed
-
- Boskey AL, Maresca M, Ullrich W, Doty SB, Butler WT, Prince CW. Osteopontin-hydroxyapatite interactions in vitro: inhibition of hydroxyapatite formation and growth in a gelatin gel. Bone Miner. 1993;22:147–159. - PubMed
-
- Boskey AL, Spevak L, Paschalis E, Doty SB, McKee MD. Osteopontin deficiency increases mineral content and mineral crystallinity in mouse bone. Calcif Tissue Int. 2002;71:145–154. - PubMed
-
- Butler WT, Bhown M, Brunn JC, D'Souza RN, Farach-Carson MC, Happonen RP, Schrohenloher RE, Seyer JM, Somerman MJ, Foster RA. Isolation, characterization and immunolocalization of a 53-kDal dentin sialoprotein (DSP) Matrix. 1992;12:343–351. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

