Mechanisms for initiation of reentry in acute regional ischemia phase 1B
- PMID: 20097623
- PMCID: PMC2829364
- DOI: 10.1016/j.hrthm.2009.11.014
Mechanisms for initiation of reentry in acute regional ischemia phase 1B
Abstract
Background: During phase 1B of acute regional ischemia, the subepicardial and subendocardial layers coupled to the inexcitable midmyocardium remain viable.
Objective: The purpose of this study was to examine how the degree of hyperkalemia in the surviving layers, the lateral width of border zone between the normal tissue and the central ischemic zone, and the degree of cellular uncoupling between the surviving layers and the midmyocardium contribute to initiation of reentry.
Methods: Simulations were conducted on the state-of-the-art model of rabbit ventricles with realistic representation of the spatial distribution of the ischemic insult.
Results: Hyperkalemia in the surviving layers led to induction of reentry by increasing refractoriness and slowing conduction in the layers. Such reentries were formed solely in the subepicardium. A minimal level of hyperkalemia was required for induction of reentry. Progress increase in hyperkalemia led to a biphasic change in vulnerability to reentry. For each level of hyperkalemia, increased cellular uncoupling between subepicardium and midmyocardium increased inducibility of reentry by restoring subepicardial tissue excitability via blocking midmyocardial electrotonic effect. In addition, increased lateral width of the border zone prevented inducibility of reentry as conduction block occurred in the central ischemic zone when the wave propagated across the border zone from the normal zone.
Conclusion: The degree of hyperkalemia in the surviving subepicardium, the lateral width of border zone, and cellular uncoupling between the subepicardium and midmyocardium determine dispersion of refractoriness, conduction velocity, excitability, and, therefore, inducibility of reentry during phase 1B.
Copyright 2010 Heart Rhythm Society. Published by Elsevier Inc. All rights reserved.
Figures



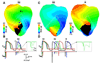
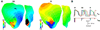
Comment in
-
Mechanisms of lethal arrhythmias due to acute myocardial ischemia: regional factors and challenges in sudden death prevention.Heart Rhythm. 2010 Mar;7(3):387-8. doi: 10.1016/j.hrthm.2010.01.001. Epub 2010 Jan 6. Heart Rhythm. 2010. PMID: 20185113 No abstract available.
Similar articles
-
Reentry in survived subepicardium coupled to depolarized and inexcitable midmyocardium: insights into arrhythmogenesis in ischemia phase 1B.Heart Rhythm. 2008 Jul;5(7):1036-44. doi: 10.1016/j.hrthm.2008.03.025. Epub 2008 Mar 25. Heart Rhythm. 2008. PMID: 18598961 Free PMC article.
-
Role of cellular uncoupling in arrhythmogenesis in ischemia phase 1B.Conf Proc IEEE Eng Med Biol Soc. 2006;2006:2272-5. doi: 10.1109/IEMBS.2006.259470. Conf Proc IEEE Eng Med Biol Soc. 2006. PMID: 17945702
-
In silico ischaemia-induced reentry at the Purkinje-ventricle interface.Europace. 2014 Mar;16(3):444-51. doi: 10.1093/europace/eut386. Europace. 2014. PMID: 24569899
-
Generation of arrhythmias in myocardial ischemia and infarction.Am J Cardiol. 1988 Jan 15;61(2):20A-26A. doi: 10.1016/0002-9149(88)90737-0. Am J Cardiol. 1988. PMID: 2447770 Review.
-
Electrophysiologic basis for arrhythmias in ischemic heart disease.Am J Cardiol. 1984 Feb 27;53(5):1B-7B. doi: 10.1016/0002-9149(84)90493-4. Am J Cardiol. 1984. PMID: 6364766 Review.
Cited by
-
Computational rabbit models to investigate the initiation, perpetuation, and termination of ventricular arrhythmia.Prog Biophys Mol Biol. 2016 Jul;121(2):185-94. doi: 10.1016/j.pbiomolbio.2016.06.004. Epub 2016 Jun 19. Prog Biophys Mol Biol. 2016. PMID: 27334789 Free PMC article. Review.
-
Current applications of mathematical models of the interstitial cells of Cajal in the gastrointestinal tract.WIREs Mech Dis. 2021 Mar;13(2):e1507. doi: 10.1002/wsbm.1507. Epub 2020 Oct 7. WIREs Mech Dis. 2021. PMID: 33026190 Free PMC article. Review.
-
Comparison of Electric- and Magnetic-Cardiograms Produced by Myocardial Ischemia in Models of the Human Ventricle and Torso.PLoS One. 2016 Aug 24;11(8):e0160999. doi: 10.1371/journal.pone.0160999. eCollection 2016. PLoS One. 2016. PMID: 27556808 Free PMC article.
-
Computational approaches to understand cardiac electrophysiology and arrhythmias.Am J Physiol Heart Circ Physiol. 2012 Oct 1;303(7):H766-83. doi: 10.1152/ajpheart.01081.2011. Epub 2012 Aug 10. Am J Physiol Heart Circ Physiol. 2012. PMID: 22886409 Free PMC article. Review.
-
Mechanisms Underlying the Emergence of Post-acidosis Arrhythmia at the Tissue Level: A Theoretical Study.Front Physiol. 2017 Mar 30;8:195. doi: 10.3389/fphys.2017.00195. eCollection 2017. Front Physiol. 2017. PMID: 28424631 Free PMC article.
References
-
- Huikuri HV, Castellanos A, Myerburg RJ. Sudden death due to cardiac arrhythmias. N. Engl. J. Med. 2001;345:1473–1148. - PubMed
-
- Kaplinsky E, Ogawa S, Balke C, et al. Two periods of early ventricular arrhythmia in the canine acute myocardial infarction model. Circulation. 1979;60:397–403. - PubMed
-
- de Groot JR, Wilms-Schopman FJ, Opthof T, et al. Late ventricular arrhythmias during acute regional ischemia in the isolated blood perfused pig heart. Role of electrical cellular coupling. Cardiovasc Res. 2001;50:362–372. - PubMed
-
- Fujiwara H, Ashraf M, Sato S, et al. Transmural cellular damage and blood flow distribution in early ischemia in pig hearts. Circ Res. 1982;51:683–693. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

