Rac1-mediated NADPH oxidase release of O2- regulates epithelial sodium channel activity in the alveolar epithelium
- PMID: 20097736
- PMCID: PMC2853346
- DOI: 10.1152/ajplung.00230.2009
Rac1-mediated NADPH oxidase release of O2- regulates epithelial sodium channel activity in the alveolar epithelium
Abstract
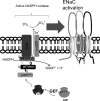
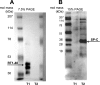
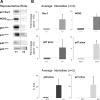
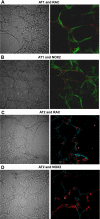
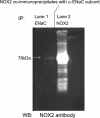
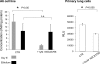
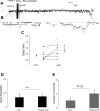
We examine whether alveolar cells can control release of O(2)(-) through regulated NADPH oxidase (NOX) 2 (NOX2) activity to maintain lung fluid homeostasis. Using FACS to purify alveolar epithelial cells, we show that type 1 cells robustly express each of the critical NOX components that catalyze the production of O(2)(-) (NOX2 or gp91(phox), p22(phox), p67(phox), p47(phox), and p40(phox) subunits) as well as Rac1 at substantially higher levels than type 2 cells. Immunohistochemical labeling of lung tissue shows that Rac1 expression is cytoplasmic and resides near the apical surface of type 1 cells, whereas NOX2 coimmunoprecipitates with epithelial sodium channel (ENaC). Since Rac1 is a known regulator of NOX2, and hence O(2)(-) release, we tested whether inhibition or activation of Rac1 influenced ENaC activity. Indeed, 1 microM NSC23766 inhibition of Rac1 decreased O(2)(-) output in lung cells and significantly decreased ENaC activity from 0.87 +/- 0.16 to 0.52 +/- 0.16 [mean number of channels (N) and single-channel open probability (P(o)) (NP(o)) +/- SE, n = 6; P < 0.05] in type 2 cells. NSC23766 (10 microM) decreased ENaC NP(o) from 1.16 +/- 0.27 to 0.38 +/- 0.10 (n = 6 in type 1 cells). Conversely, 10 ng/ml EGF (a known stimulator of both Rac1 and O(2)(-) release) increased ENaC NP(o) values in both type 1 and 2 cells. NP(o) values increased from 0.48 +/- 0.21 to 0.91 +/- 0.28 in type 2 cells (P < 0.05; n = 10). In type 1 cells, ENaC activity also significantly increased from 0.40 +/- 0.15 to 0.60 +/- 0.23 following EGF treatment (n = 7). Sequestering O(2)(-) using 2,2,6,6-tetramethylpiperidine-N-oxyl (TEMPO) compound prevented EGF activation of ENaC in both type 1 and 2 cells. In conclusion, we report that Rac1-mediated NOX2 activity is an important component in O(2)(-) regulation of ENaC.
Figures




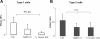
Similar articles
-
Nadph oxidase regulates alveolar epithelial sodium channel activity and lung fluid balance in vivo via O⁻₂ signaling.Am J Physiol Lung Cell Mol Physiol. 2012 Feb 15;302(4):L410-9. doi: 10.1152/ajplung.00260.2011. Epub 2011 Dec 9. Am J Physiol Lung Cell Mol Physiol. 2012. PMID: 22160304 Free PMC article.
-
Receptor for advanced glycation end-products regulates lung fluid balance via protein kinase C-gp91(phox) signaling to epithelial sodium channels.Am J Respir Cell Mol Biol. 2015 Jan;52(1):75-87. doi: 10.1165/rcmb.2014-0002OC. Am J Respir Cell Mol Biol. 2015. PMID: 24978055 Free PMC article.
-
Ethanol alters alveolar fluid balance via Nadph oxidase (NOX) signaling to epithelial sodium channels (ENaC) in the lung.PLoS One. 2013;8(1):e54750. doi: 10.1371/journal.pone.0054750. Epub 2013 Jan 29. PLoS One. 2013. PMID: 23382956 Free PMC article.
-
Role of the small GTPase Rac in p22phox-dependent NADPH oxidases.Biochimie. 2007 Sep;89(9):1133-44. doi: 10.1016/j.biochi.2007.05.003. Epub 2007 May 17. Biochimie. 2007. PMID: 17583407 Review.
-
NADPH oxidase activation in neutrophils: Role of the phosphorylation of its subunits.Eur J Clin Invest. 2018 Nov;48 Suppl 2:e12951. doi: 10.1111/eci.12951. Epub 2018 Jun 3. Eur J Clin Invest. 2018. PMID: 29757466 Review.
Cited by
-
Rational design of small molecule inhibitors targeting the Rac GTPase-p67(phox) signaling axis in inflammation.Chem Biol. 2012 Feb 24;19(2):228-42. doi: 10.1016/j.chembiol.2011.12.017. Chem Biol. 2012. PMID: 22365606 Free PMC article.
-
Intoxicated Na(+) channels. Focus on "ethanol stimulates epithelial sodium channels by elevating reactive oxygen species".Am J Physiol Cell Physiol. 2012 Dec 1;303(11):C1125-6. doi: 10.1152/ajpcell.00301.2012. Epub 2012 Sep 19. Am J Physiol Cell Physiol. 2012. PMID: 22992679 Free PMC article. No abstract available.
-
ENaCs and ASICs as therapeutic targets.Am J Physiol Cell Physiol. 2012 Apr 1;302(7):C943-65. doi: 10.1152/ajpcell.00019.2012. Epub 2012 Jan 25. Am J Physiol Cell Physiol. 2012. PMID: 22277752 Free PMC article. Review.
-
Of Mycelium and Men: Inherent Human Susceptibility to Fungal Diseases.Pathogens. 2023 Mar 14;12(3):456. doi: 10.3390/pathogens12030456. Pathogens. 2023. PMID: 36986378 Free PMC article. Review.
-
Curcumin nanoparticles ameliorate ICAM-1 expression in TNF-α-treated lung epithelial cells through p47 (phox) and MAPKs/AP-1 pathways.PLoS One. 2013 May 9;8(5):e63845. doi: 10.1371/journal.pone.0063845. Print 2013. PLoS One. 2013. PMID: 23671702 Free PMC article.
References
-
- Bedard K, Krause KH. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev 87: 245–313, 2007 - PubMed
-
- Borok Z, Liebler JM, Lubman RL, Foster MJ, Zhou B, Li X, Zabski SM, Kim KJ, Crandall ED. Na transport proteins are expressed by rat alveolar epithelial type I cells. Am J Physiol Lung Cell Mol Physiol 282: L599–L608, 2002 - PubMed
-
- Borok Z, Danto SI, Lubman RL, Cao Y, Williams MC, Crandall ED. Modulation of T1α expression with alveolar epithelial cell phenotype in vitro. Am J Physiol Lung Cell Mol Physiol 275: L155–L164, 1998 - PubMed
-
- Dise RS, Frey MR, Whitehead RH, Polk DB. Epidermal growth factor stimulates Rac activation through Src and phosphatidylinositol 3-kinase to promote colonic epithelial cell migration. Am J Physiol Gastrointest Liver Physiol 294: G276–G285, 2008 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

