The role of potassium in inflammasome activation by bacteria
- PMID: 20097760
- PMCID: PMC2856258
- DOI: 10.1074/jbc.M109.067298
The role of potassium in inflammasome activation by bacteria
Abstract
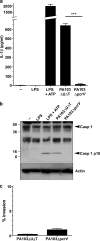
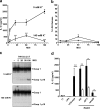
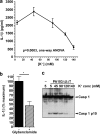
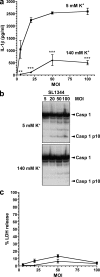
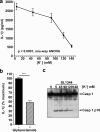
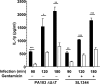
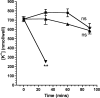
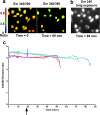
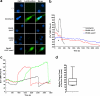
Many Gram-negative bacteria possess a type III secretion system (TTSS( paragraph sign)) that can activate the NLRC4 inflammasome, process caspase-1 and lead to secretion of mature IL-1beta. This is dependent on the presence of intracellular flagellin. Previous reports have suggested that this activation is independent of extracellular K(+) and not accompanied by leakage of K(+) from the cell, in contrast to activation of the NLRP3 inflammasome. However, non-flagellated strains of Pseudomonas aeruginosa are able to activate NLRC4, suggesting that formation of a pore in the cell membrane by the TTSS apparatus may be sufficient for inflammasome activation. Thus, we set out to determine if extracellular K(+) influenced P. aeruginosa inflammasome activation. We found that raising extracellular K(+) prevented TTSS NLRC4 activation by the non-flagellated P. aeruginosa strain PA103DeltaUDeltaT at concentrations above 90 mm, higher than those reported to inhibit NLRP3 activation. Infection was accompanied by efflux of K(+) from a minority of cells as determined using the K(+)-sensitive fluorophore PBFI, but no formation of a leaky pore. We obtained exactly the same results following infection with Salmonella typhimurium, previously described as independent of extracellular K(+). The inhibitory effect of raised extracellular K(+) on NLRC4 activation thus reflects a requirement for a decrease in intracellular K(+) for this inflammasome component as well as that described for NLRP3.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

