Endogenous morphine levels are increased in sepsis: a partial implication of neutrophils
- PMID: 20098709
- PMCID: PMC2808358
- DOI: 10.1371/journal.pone.0008791
Endogenous morphine levels are increased in sepsis: a partial implication of neutrophils
Abstract
Background: Mammalian cells synthesize morphine and the respective biosynthetic pathway has been elucidated. Human neutrophils release this alkaloid into the media after exposure to morphine precursors. However, the exact role of endogenous morphine in inflammatory processes remains unclear. We postulate that morphine is released during infection and can be determined in the serum of patients with severe infection such as sepsis.
Methodology: The presence and subcellular immunolocalization of endogenous morphine was investigated by ELISA, mass spectrometry analysis and laser confocal microscopy. Neutrophils were activated with Interleukin-8 (IL-8) or lipopolysaccharide (LPS). Morphine secretion was determined by a morphine-specific ELISA. mu opioid receptor expression was assessed with flow cytometry. Serum morphine concentrations of septic patients were determined with a morphine-specific ELISA and morphine identity was confirmed in human neutrophils and serum of septic patients by mass spectrometry analysis. The effects of the concentration of morphine found in serum of septic patients on LPS-induced release of IL-8 by human neutrophils were tested.
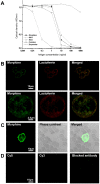
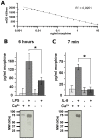
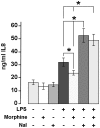
Principal findings: We confirmed the presence of morphine in human neutrophil extracts and showed its colocalisation with lactoferrin within the secondary granules of neutrophils. Morphine secretion was quantified in the supernatant of activated human polymorphonuclear neutrophils in the presence and absence of Ca(2+). LPS and IL-8 were able to induce a significant release of morphine only in presence of Ca(2+). LPS treatment increased mu opioid receptor expression on neutrophils. Low concentration of morphine (8 nM) significantly inhibited the release of IL-8 from neutrophils when coincubated with LPS. This effect was reversed by naloxone. Patients with sepsis, severe sepsis and septic shock had significant higher circulating morphine levels compared to patients with systemic inflammatory response syndrome and healthy controls. Mass spectrometry analysis showed that endogenous morphine from serum of patient with sepsis was identical to poppy-derived morphine.
Conclusions: Our results indicate that morphine concentrations are increased significantly in the serum of patients with systemic infection and that morphine is, at least in part, secreted from neutrophils during sepsis. Morphine concentrations equivalent to those found in the serum of septic patients significantly inhibited LPS-induced IL-8 secretion in neutrophils.
Conflict of interest statement
Figures



References
-
- Stefano GB, Goumon Y, Casares F, Cadet P, Fricchione GL, et al. Endogenous morphine. Trends Neurosci. 2000;23:436–442. - PubMed
-
- Gintzler AR, Gershon MD, Spector S. A nonpeptide morphine-like compound: immunocytochemical localization in the mouse brain. Science. 1978;199:447–448. - PubMed
-
- Goumon Y, Laux A, Muller A, Aunis D. Central and Peripheral Endogenous Morphine. An R Acad Nac Farm. 2009;75:389–418.
-
- Stefano GB, Kream RM. Endogenous morphine synthetic pathway preceded and gave rise to catecholamine synthesis in evolution (Review). Int J Mol Med. 2007;20:837–841. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

