Randomized controlled trial of behavioral activation smoking cessation treatment for smokers with elevated depressive symptoms
- PMID: 20099950
- PMCID: PMC3108050
- DOI: 10.1037/a0017939
Randomized controlled trial of behavioral activation smoking cessation treatment for smokers with elevated depressive symptoms
Abstract
Objective: Depressive symptoms are associated with poor smoking cessation outcomes, and there remains continued interest in behavioral interventions that simultaneously target smoking and depressive symptomatology. In this pilot study, we examined whether a behavioral activation treatment for smoking (BATS) can enhance cessation outcomes.
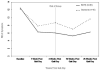
Method: A sample of 68 adult smokers with mildly elevated depressive symptoms (M = 43.8 years of age; 48.5% were women; 72.7% were African American) seeking smoking cessation treatment were randomized to receive either BATS paired with standard treatment (ST) smoking cessation strategies including nicotine replacement therapy (n = 35) or ST alone including nicotine replacement therapy (n = 33). BATS and ST were matched for contact time and included 8 sessions of group-based treatment. Quit date was assigned to occur at Session 4 for each treatment condition. Participants completed a baseline assessment; furthermore, measures of smoking cessation outcomes (7-day verified point-prevalence abstinence), depressive symptoms (Beck Depression Inventory-II; Beck, Steer, & Brown, 1996), and enjoyment from daily activities (Environmental Reward Observation Scale; Armento & Hopko, 2007) were obtained at 1, 4, 16, and 26 weeks post assigned quit date.
Results: Across the follow-ups over 26 weeks, participants in BATS reported greater smoking abstinence (adjusted odds ratio = 3.59, 95% CI [1.22, 10.53], p = .02) than did those in ST. Participants in BATS also reported a greater reduction in depressive symptoms (B = -1.99, SE = 0.86, p = .02) than did those in ST.
Conclusions: Results suggest BATS is a promising intervention that may promote smoking cessation and improve depressive symptoms among underserved smokers of diverse backgrounds.
Figures

References
-
- Armento MEA, Hopko DR. The Environmental Reward Observation Scale (EROS): Development, Validity, and Reliability. Behavior Therapy. 2007;38:107–119. - PubMed
-
- Beck AT, Steer RA, Brown G. Beck Depression Inventory II manual. San Antonio, TX: The Psychological Corporation; 1996.
-
- Cinciripini PM, Wetter DW, Fouladi RT, Blalock JA, Carter BL, Cinciripini LG, et al. The effects of depressed mood on smoking cessation: Mediation by postcessation self-efficacy. Journal of Consulting and Clinical Psychology. 2003;71:292–301. - PubMed
-
- Cook JW, Spring B, McChargue D, Hedeker D. Hedonic capacity, cigarette craving, and diminished positive mood. Nicotine & Tobacco Research. 2004;6(1):39–47. - PubMed
-
- El-Khorazaty MN, Johnson AA, Kiely M, El-Mohandes AAE, Subramanian S, Laryea HA, et al. Recruitment and retention of low-income minority women in a behavioral intervention to reduce smoking, depression, and intimate partner violence during pregnancy. BMC Public Health. 2007;7:233–250. - PMC - PubMed

